Frequency And Load Of Hepatitis B Virus Dna In First
Holger Hennig, Ines Puchta, Jurgen Luhm, Peter Schlenke, Siegfried Goerg, Holger Kirchner Frequency and load of hepatitis B virus DNA in first-time blood donors with antibodies to hepatitis B core antigen. Blood 2002 100 : 26372641. doi:
Hbcab Or The Hepatitis B Core Antibody Test
The hepatitis B core antibody is produced by your immune system after infection by the hepatitis B virus, and it can persist for life. It is a sign that you either have an new, active hepatitis B infection or that you acquired hepatitis B in the past.
HBcAb is an immune system response to a protein in the core of the virus, and it is only present if you have been infected, rather than immunized against the virus. It is part of a routine screening panel of tests for hepatitis B. If your rest results turn out to be positive, your healthcare provider will order further tests to determine the stage of the infection: acute or chronic .
Also Known As: anti-HBc, HBcAb
Detection Of Serologic Markers
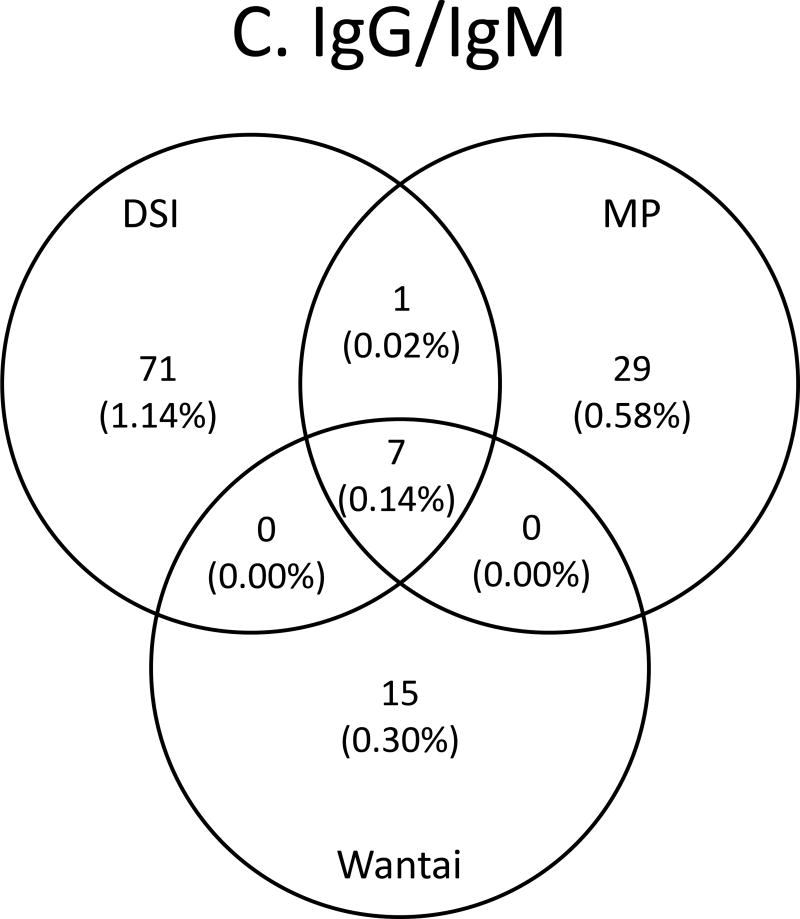
Qualitative assays for HBV serology and quantitative assay for anti-HBs, were performed using commercial kits on the automated ARCHITECT i6000 analyzer according to manufacturer’s instructions on all study samples. Both anti-HBc Total and anti-HBc IgM S/CO 1 results were considered reactive. Anti-HBs levels were expressed in International Units per liter .The assay is linear from 2 to 1,000 IU/L. Serum anti-HBs titers 10 IU/L was considered positive following WHO recommendations.
You May Like: What Kind Of Doctor Do You See For Hepatitis C
Hepatitis B Core Total Antibodies Serum
Suggests clinical disorders or settings where the test may be helpful
Diagnosis of recent or past hepatitis B infection
Determination of occult hepatitis B infection in otherwise healthy hepatitis B virus carriers with negative test results for hepatitis B surface antigen, anti-hepatitis B surface, anti-hepatitis B core IgM, hepatitis Be antigen, and anti-HBe
This assay is not useful for differentiating among acute, chronic, and past or resolved hepatitis B infection
This test is not offered as a screening or confirmatory test for blood donor specimens.
Testing Algorithm
A short description of the method used to perform the test
Chemiluminescence Immunoassay
NY State Available Indicates the status of NY State approval and if the test is orderable for NY State clients.
Lists a shorter or abbreviated version of the Published Name for a test
Lists additional common names for a test, as an aid in searching
Anti Core Antibodies
Hepatitis B Core Ab IgG and IgM, S
Hepatitis B Virus
Testing Algorithm
Describes the specimen type validated for testing
Serum SST
Date of collection is required.
Specimen Required Defines the optimal specimen required to perform the test and the preferred volume to complete testing
Collection Container/Tube: Serum gel
Collection Instructions: Centrifuge and aliquot serum into a plastic vial within 24 hours of collection.
Special Instructions
–Gastroenterology and Hepatology Client Test Request
0.4 mL
| Gross hemolysis |
| Reject |
| Specimen Type |
|---|
Can I Donate Blood After Having Hepatitis B
Is it possible to donate blood after having hepatitis B? Josh*
Hepatitis B is one of the that cause hepatitis. One way that someone can become infected with the hepatitis B virus is through blood.
People infected with hepatitis B may carry the virus without even knowing it. They can pass it to others through blood or sexual contact. Because of this, anyone who has ever tested positive for hepatitis B cannot donate blood.
It’s not just hepatitis B that affects who can donate blood. Other types of viral hepatitis, HIV, and some infections can mean that a person can’t give blood.
*Names have been changed to protect user privacy.
Read Also: How You Catch Hepatitis C
Being Tested For Hepatitis B Core Antibody
The hepatitis B core antibody test is part of a screening panel for hepatitis B, which also will include hepatitis B surface antigen and hepatitis B surface antibody . These three tests look for acute and chronic infections.
Tests may be ordered if you have symptoms of hepatitis, such as jaundice , fever, fatigue, pale stools, dark urine, nausea, vomiting, and loss of appetite. In this case, the hepatitis B core antibody IgM test may be used, as it shows an early stage of infection.
This test may be ordered if you are being screened for hepatitis B because you are donating blood or wish to become an organ donor. Hepatitis B can be transmitted by blood or through organ transplants, so donors are tested to prevent infecting recipients. It’s possible to have had the infection with only mild symptoms, so many people don’t realize they have had hepatitis B.
People who are part of populations at risk for hepatitis B infection will be screened. Screening is also often done for pregnant people, infants, people sharing a home with hepatitis B patients, people who may have been exposed by needlestick injuries or body fluids, and for people with HIV .
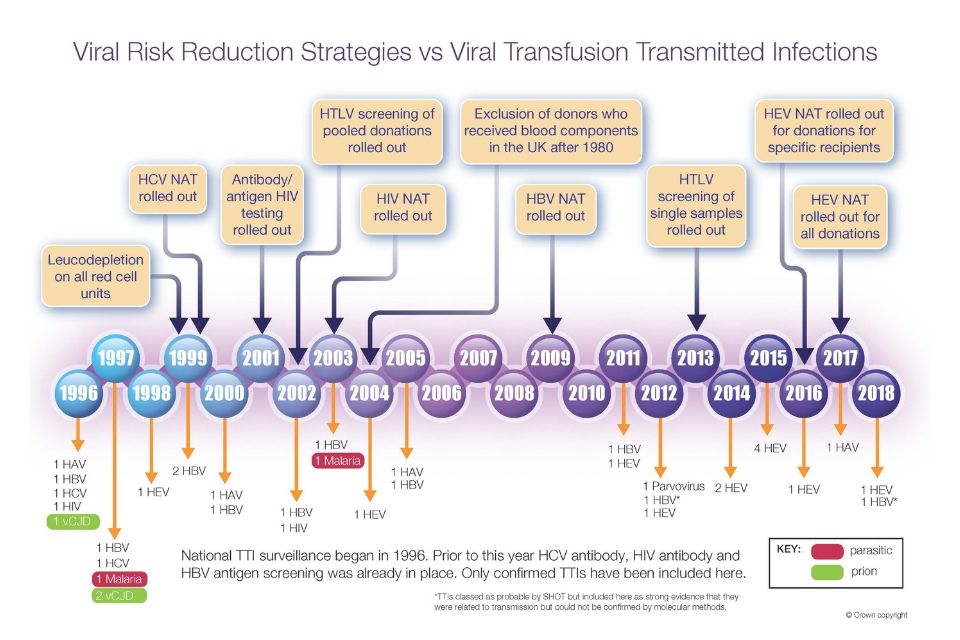
Gaps Left By Current Nat Testing
Transmission of HBV by NAT negative donations was repeatedly reported as summarized by Candotti and Laperche . Infection experiments with chimpanzees or humanized mice suggest that one single HBV particle from early phase sera may induce a full HBV infection whereas particles from later still HBsAg positive phases are usually less infectious . The recent paper from Candotti et al. provides deeper insight because it shows that in certain OBI cases HBV particles may be as infectious as particles from the early phase. Furthermore, the study illuminates the difficulties to follow-up and identify infected recipients unless the follow-up of recipients and look-back of donors with suspected donors is consequently pursued . This observation takes the minimum requirements for HBV screening procedures to a new dimension if optimum safety at all economic cost is pursued: NAT screening should be applied to all donations at a limit of 95% detection of 0.8 ge/mL which would require testing of several mL of single donations and virtually exclude minipool testing.
Don’t Miss: Where Does The Hepatitis C Virus Come From
Site Preparation And Drawing Blood
The blood is drawn from a large arm vein close to the skin, usually the median cubital vein on the inside of the elbow. The skin over the blood vessel is cleaned with an antiseptic such as iodine or chlorhexidine to prevent skin bacteria from contaminating the collected blood and also to prevent infections where the needle pierced the donor’s skin.
A largeneedle is used to minimize shearing forces that may physically damage red blood cells as they flow through the needle. A tourniquet is sometimes wrapped around the upper arm to increase the pressure of the blood in the arm veins and speed up the process. The donor may also be prompted to hold an object and squeeze it repeatedly to increase the blood flow through the vein.
The most common method is collecting the blood from the donor’s vein into a container. The amount of blood drawn varies from 200 millilitres to 550 millilitres depending on the country, but 450â500 millilitres is typical. The blood is usually stored in a flexible plastic bag that also contains sodium citrate, phosphate, dextrose, and adenine. This combination keeps the blood from clotting and preserves it during storage up to 42 days. Other chemicals are sometimes added during processing.
Apheresis is also used to collect more red blood cells than usual in a single donation and to collect white blood cells for transfusion.
Blood Screening In The United States
The U.S. Food and Drug Administration , through the Center for Biologics and Research , is responsible for ensuring the safety of the roughly 19 million units of whole blood donated in the United States each year.
To keep the blood supply safe, the FDA has established regulations to screen donors before a donation and to screen donated blood after it has been received by blood banks. To help with this, an extensive questionnaire is given to donors to collect information about their medical history and any risk factors that may exclude them from donating.
Blood received from donors then undergoes routine screening for the following blood-transmitted infections:
- Hepatitis B
- Zika virus
Any donated blood is quarantined until it is tested and shown to be free of infection.
Due to advanced blood screening practices, the risk of the accidental transmission of hepatitis B and C from contaminated blood is less than one in 500,000 and one in two million transfused units, respectively.
You May Like: Does Hepatitis C Have A Vaccine
Tests We Carry Out On Your Blood
You may have noticed that each time you give a blood donation we also take blood samples.
These samples are used to perform a range of screening tests in our laboratories.
Most of these tests are mandatory, in other words we must carry them out on every single blood donation, whether this is your first donation or just one of the many you have given over the years.
However, there are some additional tests that may need to be done on some donations as necessary.
Sometimes the tests cannot be done, for example – if you give an incomplete blood donation or no blood samples are obtained, or if we cannot take a donation because of poor veins or you have low haemoglobin level for blood donation.
The tests play a very important role in ensuring that we provide a safe blood supply to patients. We test for your blood group, so that we can select the correct group for the patient.
We also test for infections that can be passed from donor to patient via a blood transfusion.
The tests are carried out by computer-controlled automated machines which can test many samples both quickly and easily, so helping us to get blood to the hospitals as fast as we can.
Any donation that is reactive on any one of the screening tests cannot be used. If your blood is reactive on any one of the screening tests, further tests are carried out to confirm whether the result indicates a true infection.
If the test results show that you can no longer give blood, then you will be given specific advice.
Sample Collection And Preparation
Ninety-two blood donors in Ile-Ife, Osun State were enrolled for the study between July and August 2009. Five millilitres of blood was collected from each donor by venepuncture into a labelled sterile container free of anticoagulants or preservative. Each blood specimen was separated by low centrifugation at 500 g for 5 minutes and the serum transferred into labelled cryovial. Thereafter, the sera were kept frozen at -20C until analysed.
Don’t Miss: How Did I Get Hepatitis B
Blood Donor Selective Recruitment
In recent years, careful selection of blood donors became an essential and pragmatic element of blood safety management. In that respect, WHO actively promotes the recruitment of voluntary non-remunerated donors . The generally high prevalence of bloodborne pathogens observed in paid donors supported this strategy. Blood safety is improved further by encouraging VNRDs to become regular donors who show considerably lower prevalence of viral markers . This policy was successfully implemented in most of high-income countries but might have negative consequences by excluding traditional family/replacement donors that constitute 4100% of the blood supply in middle- and low-income countries , and therefore perpetuating blood shortage and increasing the cost of blood transfusion . Exclusion of FRDs relied mainly on the assumption that these donors could not be differentiated from unsafe paid donors. However, during the past few years, a wealth of evidence has been collected that showed no epidemiological and social difference between FRDs and first-time VNRDs .
Important Information About Hiv And Aids
- I am a man who has had sex with another man even one time since 1977.
- I have taken illegal drugs with a needle.
- I have taken clotting factor concentrates for a bleeding disorder such as hemophilia.
- I have taken money or drugs for sex since 1977.
- I have had sex within the last 12 months with someone who has been involved in any of the activities listed above.
- I have received blood, for any reason, within the last 12 months.
- I have had or been treated for syphilis or gonorrhea in the last 12 months.
- I have had sex in the past 12 months with someone who has AIDS or who has tested positive for the AIDS virus.
- I have been held in a correctional institution for more than 72 hours consecutively within the last 12 months.
- Fever higher than 100.5 for more than 10 days
- Unexplained sweating, especially at night
- Persistent cough
- swollen lymph nodes lasting more than one month
- White or unusual spots in your mouth that will not go away
- Blue or purple spots on or under the skin or inside the mouth or nose
- HIV antibodies
Recommended Reading: What Are The Symptoms Of Hepatitis A Virus
Hepatitis B Core Antibody
|
Organ Donor Panel 2 |
|
| Primary test for blood donor and recipient/patient testingPrimary test for organ donor testing | |
| Method | Abbott PRISM HBcore assay |
| Test Information | Assay is a chemiluminescent immunoassay for the qualitative detection of total antibody to hepatitis B core antigen in human serum and plasma specimens. |
| Sample Requirements | Serum or Plasma . Heparin anticoagulants are not acceptable. |
| Requested Volume | |
| Minimum Volume or Pediatric volume | 0.6 mL |
| Shipping Information | Samples are acceptable at 2-8°C for up to 14 days samples can be frozen at -20 °C or lower. |
| Requisition Form |
Results Of The Hbcab Test
There are two variations of antibodies. The IgM antibody is the largest antibody and the first produced in an infection. It shows that you may have a current, active infection. Sometimes it persists for years, but it usually drops to undetectable levels.
The HBcAb IgG variant is produced later in the course of the infection, and it’s likely that you will have a positive HBcAb IgG test the rest of your life.
The screening panel usually has a test that is for total HBcAb, which includes both IgM and IgG. The IgM test may be ordered to help determine if you have an acute infection.
A positive HBcAb test must be interpreted along with the results of the other tests. You may have an active or chronic infection, or you may be immune to hepatitis B due to past infection. Discuss the results with your healthcare provider. In any case, a positive HBcAb test means your blood or organs cannot be donated to a recipient.
Also Check: How Do You Know If You Have Alcoholic Hepatitis
Role Of Hbv Vaccination
Locarnini and Raimondo express in their commentary the expectation that universal hepatitis B immunization will more often protect recipients against HBV in blood donations. Even passively administered anti-HBs by concomitant donations from immunized donors protects against donations from OBI donors . The downside of vaccination is that it does not completely protect and that vaccinated donors with low or absent anti-HBs may develop after exposure an acute quasi-occult HBV infection which can only be detected by NAT. This incomplete protection occurs rarely but in increased frequency when the infecting HBV strain has a genotype different from the vaccine strain . This side effect should definitely not deter blood donation services to accept vaccinated donors with low anti-HBs. A better consequence would be that responsible institutions like pharmaceutical industry and WHO would consider the possible improvements of current HBV vaccines like use of the regional predominating HBV genotypes or preS1 containing vaccines . Unnoticed OBI of vaccinated individuals detectable by anti-HBc occurs very often in highly endemic countries. In the worst case only highly sensitive NAT can detect a recent occult HBV infection in anti-HBs positive vaccinated individuals .
Blood Groups And Blood Group Antibodies
Every donation is tested to determine the ABO and RhD group of the red cells and the plasma is screened to detect the most common blood group antibodies that might cause problems in a recipient. Some donations are tested for a wider range of clinically significantblood groups to allow closer matching and reduce the development of alloantibodies in patients who need long-term red cell transfusion support .Blood for neonatal or intrauterine use has a more extensive antibody screen .
Some group O donations are screened for high levels of anti-A and anti-B antibodies to reduce the risk of haemolytic reactions when group O plasma, platelets or other components containing a large amount of plasma are transfused to group A, B or AB patients, especially neonates and infants.
You May Like: How Do You Treat Autoimmune Hepatitis
Standard And Control Specimens
The first WHO international standard for hepatitis B virus DNA for NAT assays ,kindly provided by Dr John Saldanha, National Institute for Biological Standards and Control , Herts, United Kingdom, was used to determine the detection limit of the method and to quantify HBV DNApositive samples in international units of HBV DNA per milliliter of plasma. HBV DNApositive plasma samples from an external quality control program lyophilized and kindly provided by INSTAND were used as positive samples for the development and optimization of the method.
Recovery And Time Between Donations
Donors are usually kept at the donation site for 10â15 minutes after donating since most adverse reactions take place during or immediately after the donation. Blood centers typically provide light refreshments, such as orange juice and cookies, or a lunch allowance to help the donor recover. The needle site is covered with a bandage and the donor is directed to keep the bandage on for several hours. In hot climates, donors are advised to avoid dehydration until a few hours after donation.
Donated plasma is replaced after 2â3 days. Red blood cells are replaced by bone marrow into the circulatory system at a slower rate, on average 36 days in healthy adult males. In one study, the range was 20 to 59 days for recovery. These replacement rates are the basis of how frequently a donor can donate blood.
Plasmapheresis and plateletpheresis donors can donate much more frequently because they do not lose significant amounts of red cells. The exact rate of how often a donor can donate differs from country to country. For example, plasmapheresis donors in the United States are allowed to donate large volumes twice a week and could nominally donate 83 litres in a year, whereas the same donor in Japan may only donate every other week and could only donate about 16 litres in a year.
Read Also: Hepatic Wet Food For Cats