What About Patients With Hepatitis C Who Also Have Hepatitis B
Hepatitis B virus can flare in patients who are co-infected with hepatitis B and hepatitis C and are taking medication for hepatitis C. This has been reported as a potential risk for patients who are taking hepatitis C treatment and have underlying hepatitis B as well. The flare usually occurs within a few weeks after the patient starts taking medication for hepatitis C. Therefore, patients who have both hepatitis B and hepatitis C should be seen by a hepatitis expertbeforestarting treatment of the hepatitis C they may need to start taking hepatitis B treatment to avoid a hepatitis B flare.
Affordable Hepatitis Drug Offers New Hope To Millions
South-south drug gives people in middle-to-low-income countries access to effective treatment for disease known as the silent killer.
Kuala Lumpur, Malaysia Malaysia has registered the worlds first affordable and effective new drug for hepatitis C, offering the hope of accessible treatment to millions of people around the world at risk from a disease that has few early symptoms, is hard to diagnose and is often seen as a silent killer.
The drug, ravidasvir, was approved for use with an existing drug, sofosbuvir, in June, five years after the Malaysian government, partnered with the Drugs for Neglected Diseases initiative , a collaborative, non-profit drug research organisation to develop the drug.
We decided to work with middle-income countries to try to develop an effective treatment, said Jean-Michel Piedagnel, director of DNDi Southeast Asia. We started the clinical trial in Malaysia and Thailand saying we are also going to put on the market an affordable treatment.
The new drug is a direct-acting antiviral developed with Pharco, an Egyptian generic drug manufacturer, and is an effort to bring more competition into a market dominated by the worlds biggest pharmaceutical companies. Sofosbuvir the first DAA secured approval in the United States in 2013.
For years, the disease was treated with an array of drugs that carried debilitating side effects and often made people feel even worse.
Among them was 49-year-old Ng Song Ping.
Hepatitis C Virus Epidemiology
The World Health Organization estimates that there are 130 to 150 million cases of HCV infection worldwide, totaling 3% of the worlds population, with an average of 3 to 4 million new infections occurring each year. HCV infection causes substantial morbidity, ranging from cirrhosis to hepatocellular carcinoma , liver failure, and death. HCV infection and HCC were the leading indications for liver transplantation in the United States in 2014 however, the spectrum for transplantation is expected to shift as more patients with HCV infection are identified and successfully treated. According to estimates in 2013, 3.2 million Americans have chronic HCV infection, yet only 50% of patients infected with HCV know of their viral status of those, 7% to 11% are treated, and 5% to 6% have successful clearance of the virus. Furthermore, it is predicted that the burden of cirrhosis due to HCV infection could reach 37.2% by 2020 in infected patients.
In order to identify patients infected with HCV, recommendations have been made for routine testing in people born between 1945 and 1965 in people who have injected illegal drugs, received blood transfusion or organ transplantation prior to July 1992, or received clotting factor concentrates before 1987 in patients on long-term dialysis in children born to HCV-positive mothers in health care workers who have been exposed to HCV infection and in patients with evidence of chronic liver disease.
You May Like: Hepatitis B Homeopathy Treatment In Hindi
Risk Factors For The Hcv Virus
According to data from SEIEVA , the main risk factors for the acquisition of the infection i.e. the current and / or previous use of drugs, the use of tools for aesthetics , acupuncture, tattoos, body piercing or dental care, in premises without adequate hygienic conditions or managed by unqualified personnel are mainly distributed among the population between 34 and 55 years. But in this population a very low perception of risk persists, almost always accompanied by the absence of symptoms and a risk of transmission of the infection.
Hepatitis B, drugs that boost the immune system against the virus are being studied
03 June 2021
Strong Differences Between Regions
But the Regions are now on the starting grid with strong unevenness. The figure that catches the eye is a strong delay compared to the roadmap drawn, which should see the screening activities be completed for this first phase by 2022.
We are faced with the possibility of being able to eliminate Hepatitis C, but the confusion that arises in the implementation of the decree is singular. This is a serious delay, certainly the result of the COVID-19 pandemic which continues to absorb resources and energy, but which is also explained by a series of other critical issues.
There are difficulties in coordinating the various regional figures involved in the planning and organization of screening. It is difficult to manage, in times suited to the objectives, to define and implement a plan that knows how to find regional resources to be used for training, information, reporting, and to combine with the methods of using the funds made available by the State for screening test.
Hepatitis C, the battle to knock it out in over 50s
You May Like: Hepatitis B And C Can Be Spread By
Hepatitis C And Health
How can health-care personnel avoid exposure to HCV?
Avoiding occupational exposure to blood is the primary way to prevent transmission of bloodborne illnesses among health-care personnel. To promote blood safety in the workplace, health-care personnel should consult infectious-disease control guidance from the National Institute for Occupational Safety and Health and from CDC. Depending on the medical procedure involved, Standard Precautions may include the appropriate use of personal protective equipment .
What is the risk of acquiring hepatitis C after being accidentally exposed to HCV-contaminated blood or body fluids in the workplace?
Although sharps injuries have decreased in recent decades due to improved prevention measures, they continue to occur, placing health-care personnel at risk for several bloodborne pathogens like hepatitis C. A recent analysis of several studies revealed an overall 0.2% risk for infection among those exposed to HCV-antibody-positive blood through needlestick or sharps injuries . Updated guidelines for management and treatment of hepatitis Cexternal icon are available to provide guidance for health-care personnel who become infected via exposure to contaminated blood at the workplace.
Other than needlesticks, do other exposures place health-care personnel at risk for hepatitis C?
Should HCV-infected health-care personnel be restricted in their work?
New Oral Hepatitis C Drugs: Faq
Dec. 9, 2013 — Experts call two new drugs for hepatitis C ”game changers” that they expect will dramatically improve treatment for many of the 3 million Americans with a chronic infection. Hepatitis C can lead to liver failure.
Both new pills — Olysio and Sovaldi — work better than the current treatment for hepatitis C. They cure it more often and in less time. They also have fewer side effects.
In a major advance, the drugs could eliminate the need for some patients to take interferon, which is injected and can have unpleasant, even intolerable side effects.
“Most hepatitis C patients and most physicians would like never to use interferon again,” says Henry Masur, MD, past president of the Infectious Diseases Society of America.
The FDA approved both new oral drugs in combination with other drugs, he says. “Neither can be taken alone.” The next question, he says, is “What is the best combination?”
Doctors hope — and predict — that the combinations deemed best will often exclude the need for interferon for more and more patients. But the new drugs are expected to be more expensive.
Here, three experts address questions they get from patients about the new options.
How do Olysio and Sovaldi work?
Both Olysio and Sovaldi prevent the virus from copying itself, Masur says.
What is each drug approved for?
Olysio is approved for people with genotype 1 infection in combination with the medicines ribavirin and interferon.
How effective is each drug?
Read Also: The Cost Of Hepatitis C Treatment
How Much Will It Cost Patients To Access The New Drugs
The Pharmaceutical Benefits Scheme listing means that hepatitis C patients will only pay the normal co-payment for the new drugs. The co-payment is currently worth $6.20 for patients with concessional healthcare cards and $38.30 for general patients without concessional healthcare cards, per drug, per month. For some patients needing three drugs, for example, the co-payment will therefore be $114.90 per month, or $18.60 per month concession.
More information on co-payment charges can be found on the Commonwealth Department of Healths website
A Researcher Reflects On Progress Fighting Hepatitis C And A Path Forward
The hepatitis C virus was discovered in 1989 research thats now earned a Nobel Prize.
When I began my medical career in Hong Kong in the early 1980s, I chose to focus on hepatitis B, in part because it was very common and because the hepatitis C virus had not yet been discovered. I witnessed the devastation that this virus caused cirrhosis, liver failure and liver cancer and the lack of treatments we could offer to patients.
Back then, scientists knew there was another type of hepatitis, but no one could identify it, so we called it non-A, non-B hepatitis. I would never have imagined that during the course of my career I would witness the discovery of what came to be known as hep C and the development of a cure for nearly all patients with chronic hepatitis C in 2014.
Underscoring the importance of these discoveries for global human health, this years Nobel Prize in Physiology or Medicine was awarded jointly to Harvey J. Alter, Michael Houghton and Charles M. Rice for the discovery of the hepatitis C virus.
Effective treatment for hepatitis C has become even more relevant today in light of the recent surge in new cases of hepatitis C due to rising opioid use.
Don’t Miss: Hepatitis B Treatment Antiviral Drugs
Actions For This Page
- Recent advances in antiviral treatment have led to the development of new highly effective drugs for the treatment of all types of hepatitis C.
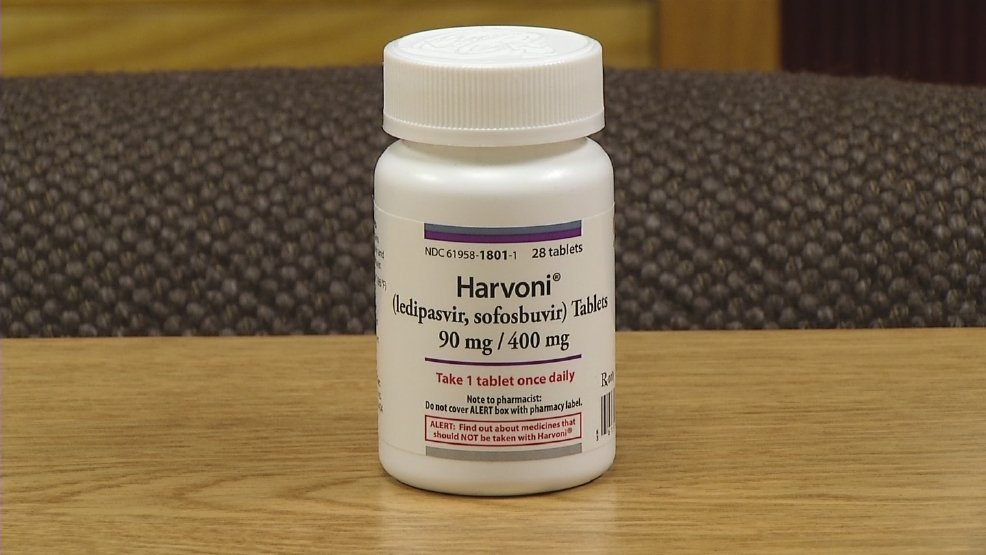
- The new hepatitis C treatments are sofosbuvir with ledipasvir sofosbuvir daclatasvir and ribavirin .
- These new treatments are now available on the Pharmaceuticals Benefits Scheme.
Are New Drugs For Hepatitis C Safe A Report Raises Concerns
- Read in app
By Denise Grady
Drugs approved in recent years that can cure hepatitis C may have severe side effects, including liver failure, a new report suggests.
The number of adverse events appears relatively small, and the findings are not conclusive. But experts said the report was a warning that should not be ignored. It involves nine widely used antiviral drugs that were heralded as a huge advance because they greatly increased cure rates, seemingly with few side effects.
The report will be published online on Wednesday by the Institute for Safe Medication Practices, a nonprofit in Horsham, Pa., that studies drug safety. Its findings are based on the groups analysis of the Food and Drug Administrations database of reports from doctors around the world of adverse events that might be related to medications.
Dr. Robert S. Brown, the director of the Center for Liver Disease and Transplantation at NewYork-Presbyterian at Columbia and Weill Cornell, who was not involved in the study, said that there had been other, scattered accounts of problems with the new drugs and that they should be investigated further.
We dont want people to ignore it and lead to risks to patients, he said. We dont want people to overreact and not treat patients who should be treated. A lot of doctors are unclear about it, and if doctors are unclear, patients are, too.
There is no vaccine to prevent the infection, so treatments have long been eagerly sought.
Also Check: How Much Is Hepatitis C Treatment
Nonstructural 5b Polymerase Inhibitors
The RdRp is vital to HCV replication, acting to catalyze RNA synthesis and genome replication. Nucleoside inhibitors arrest RNA synthesis, while nonnucleoside inhibitors bind and disrupt the RdRp function.
The nucleoside inhibitors are analogues that are incorporated into the viral RNA genome by the RdRp, causing termination of further replication, and competitively bind the active polymerase site. Single mutations can lead to resistance however, there is some evidence that mutations also seem to decrease viral fitness. This class of NS5B polymerase inhibitors has a high barrier to resistance and works broadly against genotypes with intermediate potency. Sofosbuvir was the first available NS5B nucleoside inhibitor .
Nonnucleoside inhibitors inhibit the RdRp by binding an allosteric site in a noncompetitive fashion, which changes the biochemical activity of the polymerase. They have a low barrier of resistance. Beclabuvir, an indole derivative, binds the thumb I subdomain on the RdRp with potent activity but has reduced activity against HCV genotypes 2 and 6 infections. Dasabuvir, a benzothiadiazine derivative, binds the palm I site on the RdRp, causing changes to the active site and preventing transcription., Deleobuvir and radalbuvir are additional nonnucleoside inhibitors .
What Are Genotypes And Do They Matter
Six different genotypes of hepatitis C have been identified. Genotypes 1 and 3 are the most common causes of hepatitis C in Australia and make up 90 per cent of all cases. They are important because they help determine the treatment you need. Unlike in the past, however, your genotype is not important in terms of the chance of cure. With the treatment drugs, all six genotypes have a very high chance of cure.
You May Like: What Is The Treatment For Hepatitis C Virus
What Are The New Hepatitis C Treatments And When Will They Be Available
Recent advances in antiviral treatment have led to the development of new highly effective drugs for the treatment of all types of hepatitis C.
The new hepatitis C treatments are sofosbuvir with ledipasvir sofosbuvir daclatasvir and ribavirin .
These new treatments will be available on the Pharmaceuticals Benefits Scheme from 1 March 2016.
Your Insurance Company Could Say No
Some insurance companies try to combat the high cost of hepatitis C drugs by rejecting coverage for them. More than one-third of people were denied coverage for these drugs by their insurance company, according to a 2018 study in Open Forum Infectious Diseases. Private insurance companies rejected more claims for these drugs over 52 percent than Medicare or Medicaid.
Medicare and Medicaid are more likely to approve hepatitis C drug coverage. But with Medicaid, you might have to meet certain requirements to receive these drugs, such as:
- getting a referral from a specialist
- having signs of liver scarring
- showing proof that you have stopped using alcohol or illicit drugs, if this is a problem
Don’t Miss: How Can You Get Rid Of Hepatitis C
Who Can Prescribe The New Drugs
A section 85 listing on the Pharmaceutical Benefits Scheme will allow general practitioners, as well as specialists, to prescribe the new treatments. This means that people with hepatitis C will be able to be treated by a general practitioner in the community. However, people with more advanced care needs, such as cirrhosis, may still need to see a specialist.
Why Should People Take Antiviral Medications For Hepatitis C
The purpose of taking antiviral medications for hepatitis C is to:
- remove all the hepatitis C virus from your body permanently
- stop or slow down the damage to your liver
- reduce the risk of developing cirrhosis
- reduce the risk of developing liver cancer
- reduce the risk of liver failure and the need for a liver transplant
Also Check: How Can Hepatitis C Be Treated
You May Not Need Treatment
Not everyone with hepatitis C will need to receive these expensive treatments. In up to of people with hepatitis C, the virus clears on its own within a few months without any need for medication. Your doctor will monitor you closely to see if your condition persists, and then decide if you need treatment.
A Pricey Drug And New Generics
The first combo pill with two drugs that inhibits different steps in hepatitis C replication was approved by the FDA in 2014. This pill is taken once a day for 8-12 weeks, has little to no side effects and improved the cure rate to 90-95%. It was hailed as a magical cure, but it came with a price tag of US$94,500 for a 12-week course of treatment. That led many insurers in the United States and national health departments in other countries to limit access to treatment.
Since then, several othercombo pills withsimilar cure rates that are equally well-tolerated have become available, and the cost has markedly decreased. In addition, low-cost generics and special pricing arrangements are available in many resource-limited countries.
While the current price of hepatitis C virus drugs is still very high, one needs to remember that for 95 percent of patients, this is a cure. It is unlike medicines for many illnesses that need to be taken for a long time, sometimes for the rest of the patients lives. Indeed, a cure for hepatitis C virus has allowed some patients who were on the liver transplant waiting list to reverse their liver failure, making transplantation unnecessary. This is good news not only for these patients but also for others on the waiting list.
Also Check: What Causes The Hepatitis C Virus
Is Hep C Curable
The latest drugs available for hepatitis C have high success rates when it comes to curing the condition.
In conversations with your doctor, you can discuss the full range of treatment options. Some of these are combination drugs.
But its important to note that not every medication may be effective for you, even if its for the right genotype.