How Effective Is Treatment
Direct acting antivirals cure 9 out of 10 patients with hepatitis C.
Successful treatment does not give you any protection against another hepatitis C infection. You can still catch it again.
There’s no vaccine for hepatitis C.
If treatment does not work, it may be repeated, extended, or a different combination of medicines may be tried.
Your doctor or nurse will be able to advise you.
We Have A Cure For Hepatitis C Why Are Rates So High
Renphoto via
We have had the ability to cure many people of hepatitis C — a common viral infection of the liver — since 2011. These new drugs are effective at curing most strains of the virus and have far fewer side effects than earlier generations of treatment. Despite these major scientific advances, however, hepatitis C rates remain high, particularly among those who are also living with HIV, as well as among people in prison and people using drugs, particularly drugs taken by injection.
Our lack of progress toward eliminating hepatitis lies in a poor screening and surveillance system, the continuing opioid epidemic, and the high cost of the cures — which has led many insurers, most notably Medicaid, to limit treatment to those who are already suffering some of the long-term consequences of this infection.
A Promise Of A Cure With A Cost
These new medicines are expensive. Researchers believe that as more come to the marketplace, prices will drop to stay competitive. If you have life-threatening liver damage due to the hepatitis C virus, you might be eligible for “compassionate care” rates, which can greatly reduce your out-of-pocket cost. Ask your doctor or case manager or directly contact the drug manufacturers.
Remember, there is no one-size-fits all treatment for chronic hepatitis C. Which medication is best for you depends on many things, including:
- The amount of liver damage
- Your previous treatments
You May Like: Where Can I Get Tested For Hepatitis B
What Are The Side Effects Of Treatment
Some people stop therapy because of side effects. Since hepatitis C can lead to liver damage, cirrhosis, and liver cancer if not treated, its vital to stick with a treatment plan.
Newer drugs have fewer severe side effects than pegylated interferon and ribavirin. Nevertheless, you may feel some effects while taking hepatitis C medication. Side effects can include:
- nausea, vomiting, or diarrhea
- appetite loss or weight loss
Serious side effects can occur with pegylated interferon and ribavirin treatment. If youre taking these medications, you should be monitored for these serious side effects:
- anemia
- thrombocytopenia
- light sensitivity in the eyes
- trouble breathing because of lung tissue inflammation
- suicidal thoughts, depression, or irritability
- thyroid disease
- elevated liver enzymes
- autoimmune disease flares
Some medications arent recommended if theres evidence of liver damage, like cirrhosis . A co-infection with HIV also affects medication options.
Ive Had Hep C Treatment Before But It Didnt Work What Now
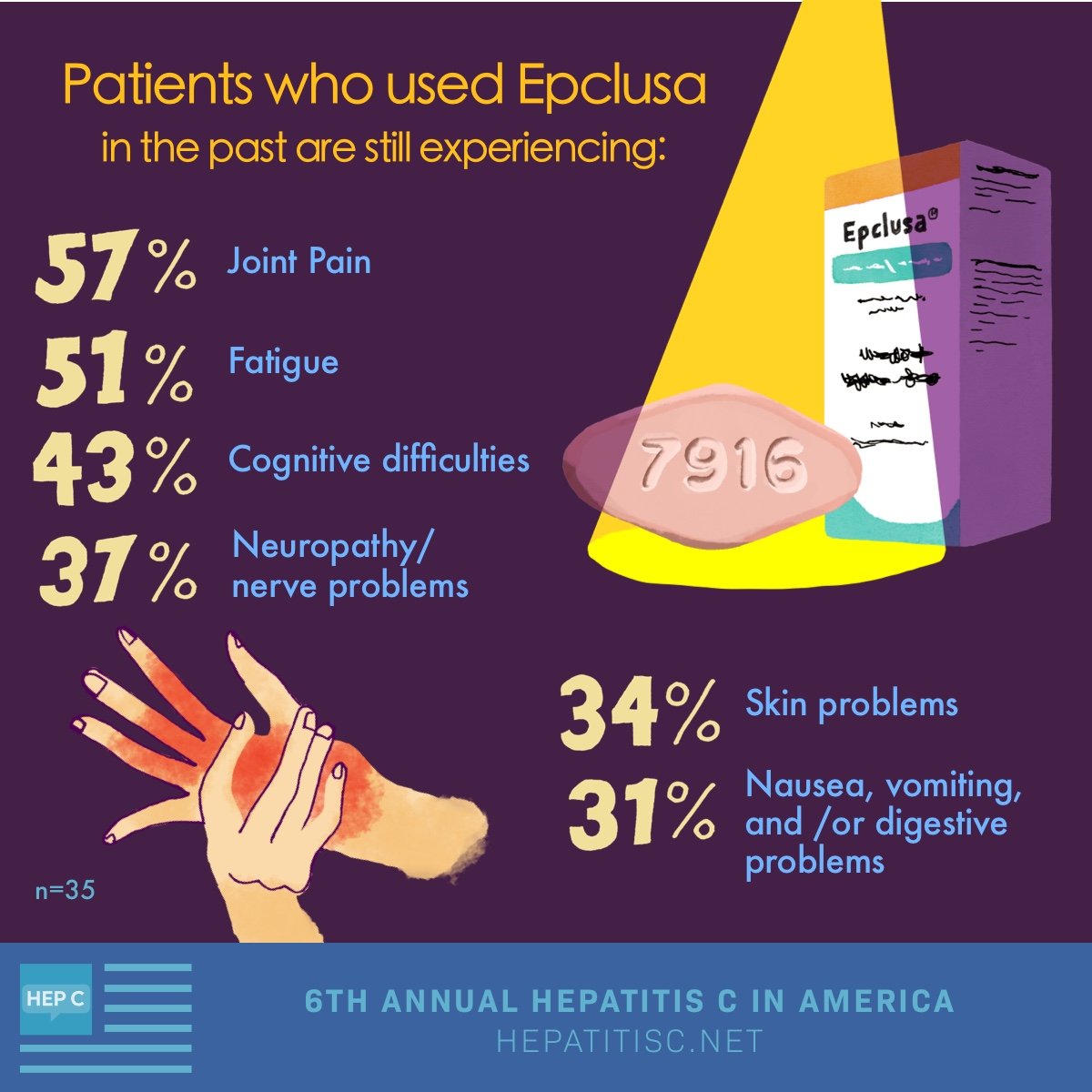
If youve had treatment for hep C before, and it didnt work, dont despair. The new treatments we talked about here still have great cure rates even if older medications didnt work for you. The choice of treatment will depend on what youve already tried, what other medications you currently take , andas alwayscost and availability.
Recommended Reading: Hepatitis A Vaccine San Diego Free
Ombitasvir Paritaprevir And Ritonavir Tablets Co
This is a relatively new group of medicines that treat genotype 1 hepatitis.
Facts about the drug pack include:
- Treatment time is 12 or 24 weeks.
- Dosage is a pack of tablets containing 12.5 mg of ombitasvir, 75 mg of paritaprevir, and 50 mg ritonavir, taken once daily in the morning, and one 250 mg tablet of dasabuvir taken twice daily with a meal.
- Common side effects of this group of drugs include nausea, itching, and trouble sleeping. If the person also takes ribavirin, side effects include tiredness, nausea, fatigue, and skin reactions.
The following medications may be effective for genotype 2:
What Does It Mean To Have A Successful Treatment What Is A Sustained Virologic Response
In an untreated state, the hepatitis C virus infects the cells of the liver and then continuously lives there, making copies of itself that circulate in the bloodstream. Antiviral medications can destroy the ability of the virus to reproduce, so the amount of virus in the bloodstream then decreases. The amount of virus in the blood is measured by aviral load.
Treatment is successful when the viral load drops toundetectablelevels, which means the virus cannot be detected in the bloodstream at all. The viral load becomes undetectable during treatment and remains undetected after treatment has ended. If there is still no detectable virus in the blood 12 weeks after the end of the treatment, the treatment was successful. This is called a Sustained Virologic Response .
A patient who has achieved an SVR is considered to be cured of the hepatitis C virus.
You May Like: Home Remedies For Hepatitis C In Urdu
Sofosbuvir Velpatasvir And Voxilapresvir
This drug combination is similar to Epclusa but also includes a drug called voxilapresvir.
Facts about Vosevi include:
- Treatment time is 12 weeks for people without cirrhosis or compensated cirrhosis .
- Dosage is fixed at 400 mg of sofosbuvir, 100 mg of velpatasvir, and 100 mg of voxilapresvir once per day with food.
- Common side effects include tiredness, a headache, diarrhea, and nausea.
Doctors often recommend Vosevi for people who have had previous treatment for hepatitis C that did not work.
Will The Drugs Be Available By 1 March 2016
Although the drugs will be available for prescribing through the Pharmaceutical Benefits Scheme, it may be that not all GPs or pharmacists are fully aware of the new treatments by that date. This means there may be some delay in some areas in accessing the drugs from your local GP. However, the Victorian Government is working with doctors, services and hospitals to ensure these delays are minimised as much as possible.
Don’t Miss: Hepatitis B Vaccine Side Effects
Can Hepatitis C Be Cured
Considerable progress has been made by past clinical trials in the medical treatment of hepatitis C. The rate of cure has increased with the development of direct-acting, all-oral antiviral regimens, and the length of therapy is much shorter. Treatment recommendations continue to change as new medicines become available. Treatment helps to reduce progression of liver damage to cirrhosis, may prevent liver cancer, and may prevent spread of the infection to other people.
Outcomes And Hcv Treatment
There is no treatment recommended for acute hep C infection however, patients should be monitored to see if the infection becomes chronic. Chronic hepatitis C can cause liver cancer or cirrhosis and is the most common reason for liver transplants in this country. The CDC estimates that of every 100 people infected with HCV, 75 to 85 will develop chronic infection, and 10 to 20 will develop cirrhosis within 20 or 30 years. Of 100 people who have hepatitis C and have developed cirrhosis, between three and six will develop liver failure each year and between one and five will develop liver cancer each year. There were 18,153 deaths related to HCV reported to the CDC in 2016, but this is likely an underestimate.
A whole new class of medication, called direct-acting antivirals , was introduced in 2011. The first two drugs to be introduced — boceprevir and telaprevir — improved the outcomes dramatically but still required patients to take interferon. About 70% of patients achieved sustained virologic response on the first generation of DAAs.
Then in 2014, Gilead Sciences introduced ledipasvir/sofosbuvir , which had a 99% SVR rate with a 12-week regimen and did not need to be combined with interferon injections. Newer DAAs have been introduced since that time, including sofosbuvir/velpatasvir and sofosbuvir/velpatasvir/voxilaprevir , also made by Gilead, elbasvir/grazoprevir by Merck, and glecaprevir/pibrentasvir from AbbVie.
Don’t Miss: Can Chronic Hepatitis B Be Cured
Nonstructural 5a Complex Inhibitors
The NS5A complex plays a role in HCV RNA replication regulation as well as viral assembly and packaging, and directly interacts with the RNA-dependent RNA polymerase . The exact antiviral action of NS5A inhibitors is unknown they are theorized to inhibit hyperphosphorylation of the NS5A protein and alter the proteins location from the endoplasmic reticulum, likely causing faulty HCV assembly. Ledipasvir, ombitasvir, daclatasvir , elbasvir, velpatasvir, odalasvir, samatasvir, ravidasvir, ruzasvir, and pibrentasvir currently make up the class of NS5A inhibitors . Ledipasvir is one of the most potent inhibitors of the NS5A complex, but may have lower activity in HCV genotypes 2 and 3 infections.,, Ombitasvir is approved in combination with paritaprevir, ritonavir, and dasabuvir as part of the 3D regimen for the treatment of HCV genotypes 1 and 4 infections, but also has a higher pill burden, which could affect compliance. Velpatasvir has antiviral activity against HCV replicons in genotypes 1 through 6. NS5A complex inhibitors have high potency, multigenotypic coverage, and generally a low barrier to resistance. Newer agents in this class have the promise to increase the resistance threshold.
Medical Treatment For Hepatitis A B & C
Treatment for hepatitis A, B, or C is based on which type of hepatitis is present in the bloodstream and the severity of the resulting liver damage. Depending on the results of diagnostic tests, our specialists at NYU Langone may recommend antiviral medication to stop the virus from replicating and protect your liver from further damage.
You May Like: Symptoms Of Advanced Hepatitis C
What Is Hepatitis C
Hepatitis C, or hep C, is an infection of the liver caused by the hepatitis C virus . The virus is transmitted through the blood, most commonly through contaminated needles, but also through sex. In 75% to 85% of cases, the infection becomes chronic, which means the body cannot get rid of it. Chronic hepatitis C can slowly destroy the liver over several decades by causing liver cirrhosis and liver failure. It can also cause a type of liver cancer called hepatocellular carcinoma.
In the US, Latin America and Europe, the most common type of hepatitis C infection is type 1.
Here, well talk about treatment options for chronic HCV type 1, which makes up 60% to 75% of US cases of hepatitis C.
How Is Hepatitis C Treated
Hepatitis C virus is treated with all-oral medications. These pills, calledantiviral medications, are usually taken once per day. These antiviral medications are extremely good at attacking the virus and preventing it from multiplying.
Antiviral medications were not the original treatment for hepatitis C. Before 2014, the only treatment for hepatitis C was called interferon and ribavirin, taken as weekly injections under the skin, plus pills. Interferon treatment caused many unpleasant side effects and was not usually successful. Then a new generation of medications became available. These antiviral treatments are extremely successful at curing the virus and have very minimal side effects.
Ribavirin is still sometimes prescribed to be taken along with the new antiviral medicines, but it has become more and more uncommon that ribavirin is needed at all. Ribavirin has some mild-moderate side effects. Ribavirin is a pill taken twice per day, as 2 or 3 pills in the morning plus 2 or 3 pills at night, depending on the patient’s body weight. Most patients do not need ribavirin.
Read Also: How Contagious Is Hepatitis C
Will A Specialist Need To Be Involved
In order to prescribe, general practitioners including physicians with expertise in viral hepatitis, will be required to first consult with a gastroenterologist, hepatologist or infectious diseases physician to ensure patients with liver disease or other complex needs are appropriately referred to specialist care. A face to face consult with the specialist is not required and patients with complex needs will likely be referred to specialist care where appropriate.
Patients affected by hepatitis C with severe or advanced liver disease may still need to access the treatments under the care of a specialist – such as a gastroenterologist, hepatologist, or an infectious disease physician with experience in treating chronic hepatitis C infection.
Fda Approves Breakthrough New Drug For Hepatitis C
The CDC estimates 3.2 million people have hepatitis C and 800,000 don’t know they have it.
A new drug to treat hepatitis C that’s been hailed as a major breakthrough received FDA approval on Friday, just two weeks after another much-anticipated hepatitis C drug was approved. Sofosbuvir is a nucleotide analog inhibitor that blocks a protein the hepatitis C virus needs to replicate.
Sovaldi, made by Northern California-based Gilead Sciences, is the first drug the FDA has approved to treat hepatitis C without the addition of interferon, a drug that must be injected and can cause debilitating side effects. For the many patients who can’t tolerate interferon treatment, Sovaldi – taken as a once-a-day pill – is the breakthrough they’ve been waiting for. Sovaldi is also approved to treat four different genotypes, or strains, of hepatitis C.
The FDA approved Sovaldi as part of combination antiviral therapy with either ribavarin alone, or ribavarin and interferon. In the FDA’s announcement, Edward Cox, MD, director of the Office of Antimicrobial Products, called sofosbuvir “a significant shift in the treatment paradigm.”
Without interferon, the only common side effects reported for Sovaldi were fatigue and headache. Combination antiviral treatment including peginterferon-alfa caused the typical more serious side effects including nausea, anemia, insomnia, and flu-like symptoms.
Drugs Needed By Millions
Two Weeks, Two Breakthrough Treatments
A Battle Over Cost?
Read Also: How Do You Get Hepatitis A And B
A Researcher Reflects On Progress Fighting Hepatitis C And A Path Forward
The hepatitis C virus was discovered in 1989 research thats now earned a Nobel Prize.
When I began my medical career in Hong Kong in the early 1980s, I chose to focus on hepatitis B, in part because it was very common and because the hepatitis C virus had not yet been discovered. I witnessed the devastation that this virus caused cirrhosis, liver failure and liver cancer and the lack of treatments we could offer to patients.
Back then, scientists knew there was another type of hepatitis, but no one could identify it, so we called it non-A, non-B hepatitis. I would never have imagined that during the course of my career I would witness the discovery of what came to be known as hep C and the development of a cure for nearly all patients with chronic hepatitis C in 2014.
Underscoring the importance of these discoveries for global human health, this years Nobel Prize in Physiology or Medicine was awarded jointly to Harvey J. Alter, Michael Houghton and Charles M. Rice for the discovery of the hepatitis C virus.
Effective treatment for hepatitis C has become even more relevant today in light of the recent surge in new cases of hepatitis C due to rising opioid use.
Actions For This Page
- Recent advances in antiviral treatment have led to the development of new highly effective drugs for the treatment of all types of hepatitis C.
- The new hepatitis C treatments are sofosbuvir with ledipasvir sofosbuvir daclatasvir and ribavirin .
- These new treatments are now available on the Pharmaceuticals Benefits Scheme.
Don’t Miss: What Does Non Reactive Hepatitis B Mean
What Are Genotypes And Do They Matter
Six different genotypes of hepatitis C have been identified. Genotypes 1 and 3 are the most common causes of hepatitis C in Australia and make up 90 per cent of all cases. They are important because they help determine the treatment you need. Unlike in the past, however, your genotype is not important in terms of the chance of cure. With the treatment drugs, all six genotypes have a very high chance of cure.
Are There Ways To Cure Hepatitis C Other Than With Medications
Patients sometimes ask whether there are ways to treat hepatitis C other than taking medicines. Currently, there are no vaccines to prevent hepatitis C. Once a person is infected, the only way to treat it is with prescribed antiviral medications.
Some patients worry that having hepatitis C means they will need a liver transplant. Only a very small fraction of people with hepatitis C require a liver transplant. By far, most people with hepatitis C never need a liver transplant. A transplant is performedonlywhen damage to the liver is extremely advanced and the liver is unable to perform its basic functions. A transplant provides a new working liver, but a transplant does not get rid of the hepatitis C virus in the patient. Patients with a liver transplant still need antiviral medication to cure their virus.
Also Check: How Does One Get Hepatitis C
Fda Approves New Drug To Treat Hepatitis C
U.S. regulators have approved the first drug to treat all forms of hepatitis C in as little as eight weeks.
The pill combination from AbbVie Inc. was approved Thursday by the Food and Drug Administration for adults without significant cirrhosis, a type of liver disease, and many patients who were not cured by prior treatment.
Mavyret joins two other AbbVie hepatitis C drugs, one from Merck & Co. and four from Gilead Sciences Inc. on the market. That gives doctors and patients more options, and insurers more leverage to wring discounts out of companies to cover the pricey drugs.
An estimated 2.7 million to 3.9 million Americans have hepatitis C, according to the Centers for Disease Control and Prevention. It usually develops slowly over decades, with no symptoms until serious damage is done. Without treatment, the virus can cause liver failure and liver cancer, often requiring a liver transplant or resulting in death.
For decades, hepatitis C treatment required a year of grueling shots and pills that gave patients flu-like symptoms and still barely cured half of them. Starting late in 2013, Gilead revolutionized treatment with the first pill-only medicines that brought cures in 12 weeks for more than 90 percent of patients.
Abbvie, based in North Chicago, Illinois, said Mavyret’s list price without insurance will be $26,400 for eight weeks’ treatment, $39,600 for 12 weeks’ treatment and $52,800 for 16 weeks’ treatment.