What Laboratory Studies Should Be Ordered To Help Establish The Diagnosis How Should The Results Be Interpreted
Laboratory studies rarely contribute to the diagnosis of the PVT, but may assist in identifying risk factors for and causes of PVT. In cirrhotic PVT, there will be abnormal INR, APTT, pancytopenia, and hyperbilirubinemia. In non-cirrhotic PVT, there may be elevation of lipase and amylase in the setting of acute pancreatitis, and leukocytosis and positive blood cultures point to the possibility of pylephlebitis. There may be a mild transaminitis in the setting of PVT. Elevated lactate may point toward concomitant mesenteric venous infarction, which may indicate the need for urgent surgical evaluation if consistent with the clinical presentation.
If there is concern for a hypercoagulable disorder, consider checking as an outpatient the following studies: antiphospholipid antibody, beta-glycoprotein, protein C & S, etc. Outpatient hematology referral is important upon discharge.
I What Every Physician Needs To Know
Portal vein thrombosis is the occlusion of the portal vein by a thrombus. The portal vein is a vital vessel that provides up to 75% of blood supply to the liver. While PVT occurs in both the pediatric and adult populations, this chapter will focus on the adult patient.
Acute and chronic PVT are considered distinct clinical entities although they represent the progression of the same disease state. Acute PVT represents an abrupt total or partial occlusion of the portal vein, and patients may present with no symptoms, or with abdominal pain, nausea and/or vomiting, diarrhea, low back pain, and fever in the setting of pylephlebitis . Chronic PVT may present with sequelae of portal hypertension, most commonly esophageal, and gastric variceal bleeding in the acute hospital setting, although hypersplenism and ascites are also seen.
Predisposing factors for PVT include stasis of hepatic venous blood flow, vessel wall damage, and hypercoagulability . In adults, the most common causes of PVT can be divided into localized or systemic etiologies . Approximately one-fourth of cases have no identifiable cause.
A History Part I: Pattern Recognition Potential Causes Of Portal Vein Thrombosis Can Be Classified As Localized Or Systemic
Localized: cirrhosis malignancy hepatocellular carcinoma, gastric and pancreatic adenocarcinoma abdominal infection abdominal inflammatory conditions pancreatitis, cholecystitis, inflammatory bowel disease, injury to portal vein due to surgery or trauma.
Systemic: hypercoagulable states, such as antiphospholipid antibody syndrome prothrombin G20210A gene mutation, hyperhomocysteinemia, myeloproliferative disease, such as JAK2V617f mutation, and protein C or S deficiency.
These potential causes of PVT can be further classified into three large groups, which is important to acknowledge for treatment discussion: 1) Cirrhotic-related PVT in the setting of underlying liver disease or cirrhosis 2) Malignancy-related PVT, i.e., in hepatocellular carcinoma, and gastric and pancreatic adenoma 3) Non-cirrhotic PVT, i.e., a genetic hypercoagulable condition, antiphospholipid antibody syndrome, and myeloproliferative disease.
In many cases, the symptoms of acute PVT are masked by the precipitating disease process , and PVT is frequently discovered incidentally on radiographic imaging. Acute PVT may be marked by abdominal pain, nausea, and/or vomiting, low back pain, and fever in the setting of septic portal vein thrombus .
Severe abdominal pain and hematochezia may indicate intestinal infarction secondary to associated mesenteric venous thrombosis.
You May Like: What Are The Early Symptoms Of Hepatitis C
Superior Vena Cava Thrombosis
Obstruction of the superior vena cava can be caused by malignant or benign disease. Malignancy is the commonest cause of SVC thrombosis accounting for up to 60% cases . Non-malignant causes are frequently due to indwelling central venous access devices or to pacemaker wires . Other recognized causes include infection and mediastinal fibrosis .
The most frequent presenting signs and symptoms of SVC thrombosis are face or neck swelling, upper limb swelling, dyspnoea, cough and dilated chest vein collaterals . The prevalence rate is unclear. However in the USA it is reported in 1â3% of cases of central venous access devices and 0·2â3·3% of cases of implanted pacemakers . CT scanning is the first imaging usually performed and contrast venography is performed if endovascular intervention is planned.
A Core Indicator Standards And Documentation
The Joint Commission has issued a National Patient Safety Goal with the goal to reduce the likelihood of patient harm associated with anticoagulant therapy. In order to meet the standard, a hospital must fulfill the following elements of performance:
Use approved protocols for the initiation and maintenance of anticoagulant therapy.
Before starting a patient on warfarin, assess the patients baseline coagulation status for all patients receiving warfarin therapy, use a current INR to adjust this therapy. The baseline status and current INR are documented in the medical record.
Use authoritative resources to manage potential food and drug interactions for patients receiving warfarin.
When heparin is administered intravenously and continuously, use programmable pumps in order to provide consistent and accurate dosing.
A written policy addressing baseline and ongoing laboratory tests that are required for anticoagulants.
Read Also: Can You Get Hepatitis C From Drinking After Someone
Who Is At Risk For Svt
SVT has long been defined as primary or secondary, depending on the presence or absence of associated abdominal or systemic risk factors. Although such a distinction is not always clear in clinical practice, emerging risk factors have decreased the number of truly unprovoked events, such that in recent reports, they account for 15% to 27% of patients.,
The relative incidence of risk factors varies with age, economic status, geographical area, and site of thrombosis. For example, a local precipitating factor is rare in BCS, but not in PVT and MVT. Thus, hematologic disorders, autoimmune diseases, and the use of hormonal therapy are the most common risk factors in BCS, whereas liver cirrhosis, abdominal cancer, intraabdominal inflammatory conditions, and surgery are the most common risk factors in PVT/MVT. A list of risk factors associated with SVT is reported in .
Sex-specific risk factors such as oral contraceptives, hormone replacement therapy, pregnancy, and puerperium are particularly relevant to the pathogenesis of BCS,, with a reported prevalence of nearly 40% in both retrospective and prospective cohort studies., In PVT, the reported prevalence of these risk factors is lower, ranging from 4% to 15%.,
C Laboratory Tests To Monitor Response To And Adjustments In Management
INR monitoring for coumadin and factor Xa level for LMWH in the setting of anticoagulation therapy for PVT. However, it is important to acknowledge that cirrhosis affects production of both pro-coagulant and anti-coagulant and the monitoring of INR and anti-factor Xa assay may be affected.
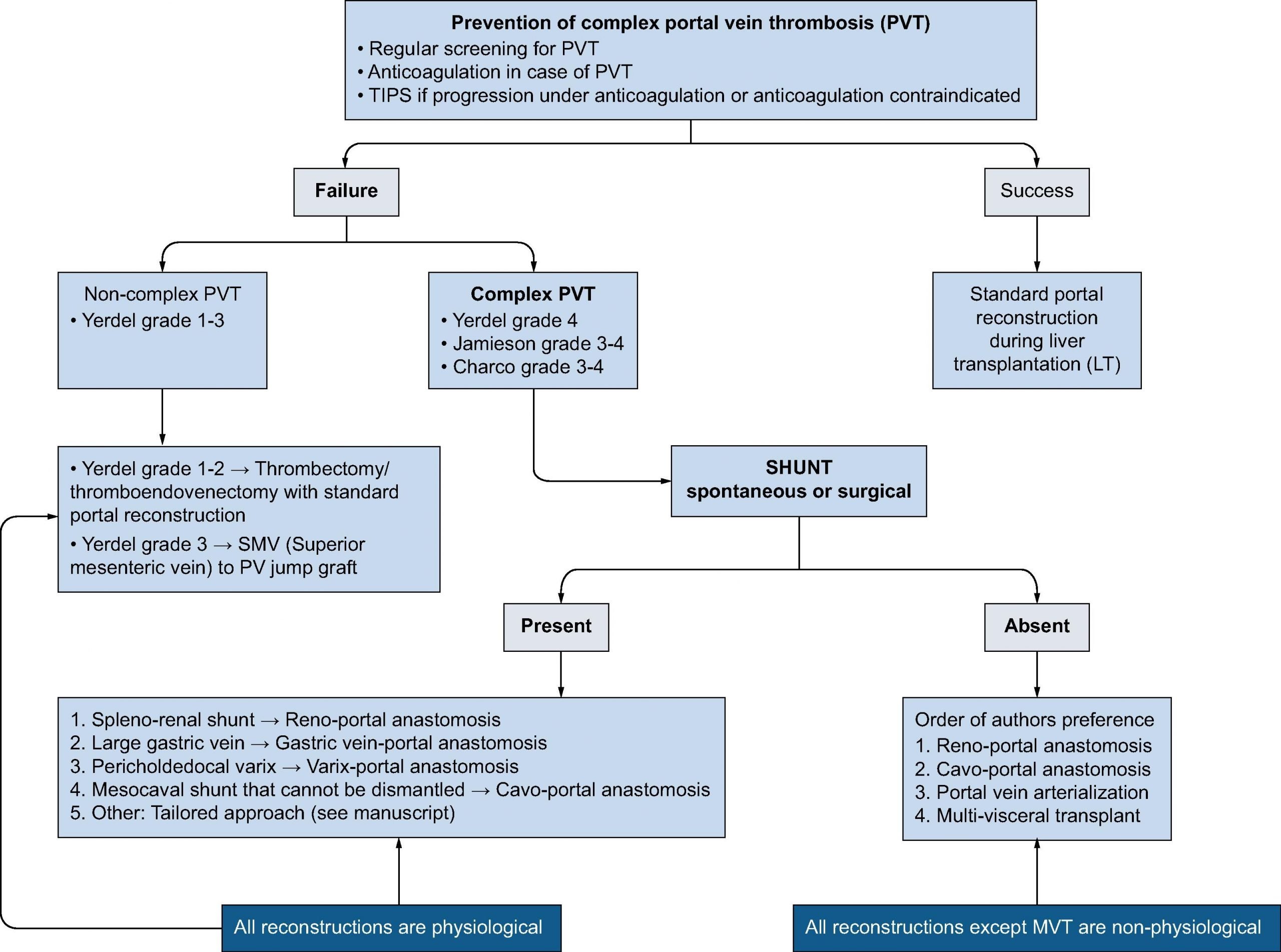
Untreated potential transplant candidates with PVT may benefit from imaging follow-up every 3 months, and consider anticoagulation in case progression.
Also Check: Is There A Cure For Hepatitis B Virus
C History Part : Competing Diagnoses That Can Mimic Portal Vein Thrombosis
Malignant invasion of the portal vein, such as in the setting of hepatocellular carcinoma, gastric carcinoma, pancreatic adenocarcinoma, and cholangiocarcinoma, can mimic thrombotic occlusion of the portal vein. For patients under consideration for liver transplantation, this distinction is critically important, as malignant invasion of the portal vein is a contraindication to transplantation.
Cirrhotic Patients Without Underlying Malignancies
Prognosis: Acute PVT occurring in cirrhotic patients without evidence of underlying malignancies constitutes 20-25% of all PVT cases. PVTs in this setting can result in both immediate and delayed complications. The impact of PVT on survival of nontransplant patients with liver cirrhosis is conflicting. In a series of 185 patients with advanced cirrhosis and bleeding esophageal varices, Amitrano et al. found that the presence of PVT was associated with nonstatistically significant predictors of 5-day mortality rate . Only Child-Pugh staging and white blood cell count demonstrated statistical significance as independent variables. It was reported that 16.8% of patients failed the protocol control of variceal bleeding and 87.1% of these cases resulted in death. In the majority of these cases, death was due to an irreversible worsening of liver function.
Scheiner and colleagues recently reported a retrospective study of 51 patients with nonmalignant cirrhosis. Twelve of these patients were given early short-term anticoagulation and thirty-nine were observed initially and then given long-term anticoagulation. The long-term anticoagulation was given for at least 9 months. The length of early anticoagulation administration was not specified and the decision to start early anticoagulation was started at the discretion of the providers. The 51-patient cohort comprised all CP stages and had a mean MELD of 13.6. The findings are:
Read Also: Hepatitis A Vaccine At Cvs
Impact Of Pvt On The Prognosis Of Liver Cirrhosis
Statement 2. PVT negatively influences patient prognosis in liver cirrhosis.
PVT may increase the risk of longterm mortality, hemorrhage, ascites, acute kidney injury, and posttransplant mortality., Therefore, the stage, grade, and extension of PVT should be considered for the assessment of patient prognosis in liver cirrhosis. But the impact of PVT on patient prognosis of liver cirrhosis may depend upon the severity of underlying liver diseases. Senzolo et al found that unrecanalized PVT after anticoagulation therapy increased the mortality of patients with ChildPugh B and C cirrhosis only. However, another study in which only patients with ChildPugh A and B cirrhosis were included showed that PVT did not increase the risk of hepatic decompensation or death. Taken together, PVT may primarily affect the prognosis of cirrhotic patients with poor liver function. Additionally, when PVT affects the mesenteric vein, liver transplantation would become more technically complicated.
Because Of The Initial Suspicion Of Cancer
Hepatic vein thrombosis treatment. Venous thrombosis within the hepatic vasculature is unique among the subtypes of deep vein thrombosis DVT. It is associated with a distinct set of predisposing factors precipitates particular morbidities and is subject to specific management considerations. As a result they suggested that.
Recurrent breast cancer was ruled out. For gradual clot growth patients may be prescribed anticoagulant. Blood count was unremarkable and Janus kinase 2 JAK2 V617F mutation was absent.
In Korea resulting from the multicenter studies hepatic resection as a first treatment option indicated the better long-term OS than TACE or sorafenib for resectable HCC patients with type I PVTT tumor thrombi involving the segmental branches of the portal vein or above and good hepatic functions. Hepatic vein angioplasty with or without stenting has been found particularly beneficial in a small subset of BCS patients with short-segment hepatic vein stenosis. Percutaneous angioplasty followed by portosystemic shunt including a transjugular intrahepatic portosystemic stent shunt and eventually liver transplantation can be proposed in a graded manner.
42 64 Manifestations of. A minority of patients can be treated medically with sodium restriction diuretics to control ascites anticoagulants such as heparin and warfarin and general symptomatic management. Resection of HCC may be attempted even in patients with IVCTT in the presence of good liver function.
Read Also: Is There A Shot For Hepatitis C
Diagnosis And Differential Diagnosis
Statement 8. PVT in liver cirrhosis can be diagnosed based on the history of chronic liver disease and typical imaging characteristics. Liver biopsy should be performed when imaging examination shows the presence of PVT but without sufficient evidence for cirrhosis. Cirrhotic PVT should be differentiated from noncirrhotic PVT and portal vein tumor thrombosis based on a combination of biochemical indicators, serum fetoprotein levels, imaging characteristics, and pathological findings.
F Prognosis And Patient Counseling
The outcomes for acute and chronic PVT are influenced primarily by the underlying disease state or associated co-morbidities. Overall mortality for acute PVT has been reported to be 10%. Patients with chronic PVT who have underlying cirrhosis and malignancy have substantially higher mortality compared to others .
Also Check: Can Hepatitis C Go Away On Its Own
Hepatic Vein Thrombosis Treatment
Budd-Chiari syndrome also known as hepatic venous outflow obstruction HVOO refers to the clinical picture that occurs when there is partial or complete obstruction of the hepatic veinsIt is characterized on imaging by ascites caudate hypertrophy peripheral atrophy and prominent collateral veins. At necropsy thrombosis of large hepatic veins was found in 6 of patients with.
Non Malignant Portal Vein Thrombosis In Liver Cirrhosis Diagnosis And Treatment European Medical Journal
What Happens During The Tips Procedure
During the TIPS procedure, a radiologist makes a tunnel through the liver with a needle, connecting the portal vein to one of the hepatic veins . A metal stent is placed in this tunnel to keep the tunnel open.
The TIPS procedure reroutes blood flow in the liver and reduces pressure in all abnormal veins, not only in the stomach and esophagus, but also in the bowel and the liver.
The TIPS procedure is not a surgical procedure. The radiologist performs the procedure within the vessels under X-ray guidance. The procedure lasts 1 to 3 hours. You should expect to stay in the hospital 1 to 2 days after the procedure.
The TIPS procedure controls bleeding immediately in over 90% of patients. However, in about 30% of patients, the shunt may narrow, causing varices to bleed again at a later time.
You May Like: How Does One Get Hepatitis B And C
What Happens During The Dsrs Procedure
The DSRS is a surgical procedure. During the surgery, the vein from the spleen is detached from the portal vein and attached to the left kidney vein. This surgery selectively reduces the pressure in your varices and controls the bleeding.
A general anesthetic is given to you before the surgery. The surgery lasts about 4 hours. You should expect to stay in the hospital from 7 to 10 days.
DSRS controls bleeding in over 90% of patients the highest risk of any recurrent bleeding is in the first month. However, the DSRS procedure provides good long-term control of bleeding.
A potential complication of the DSRS surgery is ascites . This can be treated with diuretics and restricted sodium intake.
Conflict Of Interest Statement
BK has received research support from the CSL Behring Foundation, NovoNordisk A/S, and Bayer Healthcare US and also serves on advisory boards for NovoNordisk A/S and Bayer Healthcare US. RK serves on an advisory board for Bayer Healthcare US. The authors have no actual or potential conflict of interest.
You May Like: What Is A Hepatitis Panel
Axillary Subclavian And Brachial Vein Thrombosis
Upper extremity deep vein thromboses may involve the axillary, subclavian and brachial veins. These account for up to 10% of all DVTs , and occur with a rate of around 16 per 100 000 of the population per annum . UEDVT are considered primary if they are idiopathic or associated with thoracic outlet syndrome or effort or secondary if they are associated with an underlying precipitant such as placement of a central venous catheter.
Thoracic outlet syndrome comprises compression of the neurovascular bundle in the thoracic outlet. The compression may be due to either boney structures, such as the first rib and clavicle, or muscle bulk. PagetâSchroetter syndrome is considered a form of TOS in which thrombosis is induced by microtrauma to the vessel after vigorous effort.
A systematic review of poor methodological quality studies concluded that compression ultrasound with a sensitivity of 97% may be an acceptable alternative to venography in the investigation of suspected UEDVT . The use of clinical decision rules combined with D-dimer testing for the exclusion of UEDVT has not been adequately assessed .
The data on complication rates for UEDVT are derived from heterogeneous cohorts of patients. Pulmonary embolism , post-thrombotic syndrome and annual recurrence rates of 2â8% are reported . In a direct comparison of outcomes in patients with idiopathic UEDVT and lower extremity DVT, 5-year recurrence rates of 2% and 19% respectively were described .
Who Should Be Treated
Treatment of acute SVT aims to achieve vessel recanalization and avoid complications, such as bowel ischemia or chronic portal hypertension.67 Therefore, starting anticoagulant therapy in acute symptomatic SVT without absolute contraindications is strongly recommended.2,3,69 Prevention of thrombosis progression or recurrence is the main goal of anticoagulant therapy in patients with chronic SVT, especially those with persistent risk factors. However, several areas of uncertainty still exist in the treatment of chronic SVT.67,75
You May Like: What Does Hepatitis B Come From
Risk Factors And Natural History
Since the first description of portal vein thrombosis by Balfour and Stewart in 1869, in their report of a 20-year-old patient who died from complications of splenomegaly, ascites, and varicose dilation with associated PVT, the clinical manifestations and management of PVT have been met with great interest. PVT is classically defined as obstruction of the portal vein by thrombus, graded based upon the extent of vascular occlusion. As an entity, it has been included occasionally in splanchnic vein thrombosis , referring to the thrombotic involvement of portal, mesenteric, splenic veins, and in Budd-Chiari syndrome, based on shared etiology and risk factors.
The incidence rate of PVT is unknown. The population prevalence has been reported as 1% based on an autopsy series from Sweden and has been proposed to range between 0.6% to 26% in patients with liver cirrhosis, in which the incidence of PVT rises with severity of liver disease. In patients undergoing liver transplantation for end-stage liver diseases, for example, studies have reported 5.5% to 26% prevalence.
What Imaging Studies Should Be Ordered To Help Establish The Diagnosis How Should The Results Be Interpreted
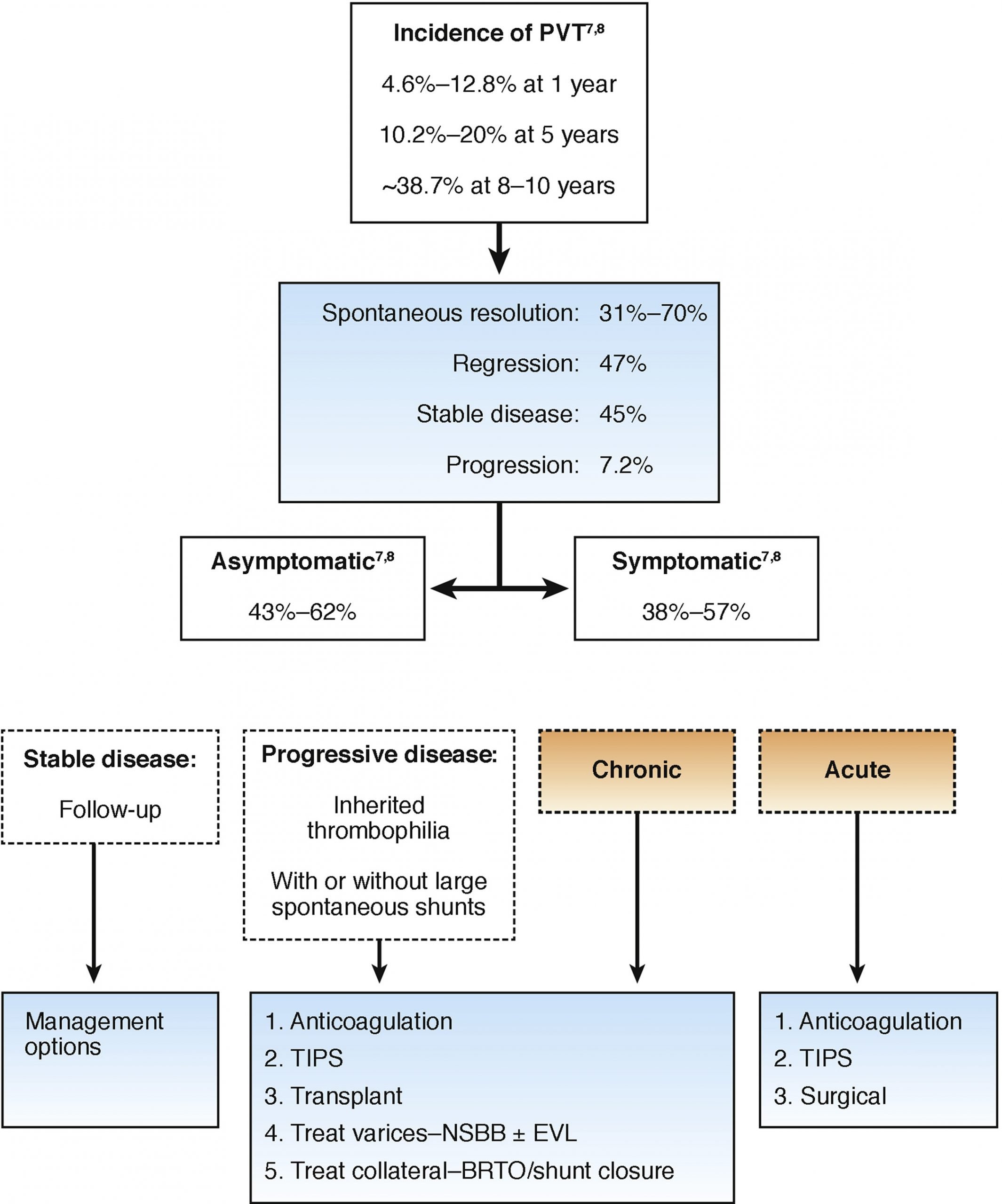
PVT is confirmed by radiographic studies, and is often discovered incidentally. Doppler ultrasound of the abdomen, computed tomography angiography, and magnetic resonance angiography are the most commonly employed modalities. Digital subtraction angiography offers the gold standard but is rarely required to establish the diagnosis.
When acute PVT is clinically suspected, the AASLD advises initial imaging with a pre- and post-contrast enhanced CT scan, or Doppler ultrasound if CT is unavailable. CT images characteristically demonstrate absence of portal vein enhancement. CT and MR angiography provide superior assessment of thrombus extension to the mesenteric veins. Imaging may also provide evidence of underlying precipitants for the PVT, such as hepatic cirrhosis, malignancy, or an intra-abdominal inflammatory process. In patients with end-stage liver disease and/or kidney impairment, caution should be taken with using contrast for further diagnosis, so ultrasound abdomen with Doppler would be the test of choice.
When evaluating for chronic PVT, the AASLD advises initial imaging with Doppler ultrasound, then contrast-enhanced CT scan or MRI. Imaging in chronic PVT may demonstrate replacement of the portal vein by a portal cavernoma: a tortuous structure of collateral veins in the absence of a visible portal vein. The hepatic arteries may be enlarged. Again, radiographic imaging may reveal potential underlying causes such as hepatic cirrhosis.
You May Like: What Is The Treatment For Hepatitis C