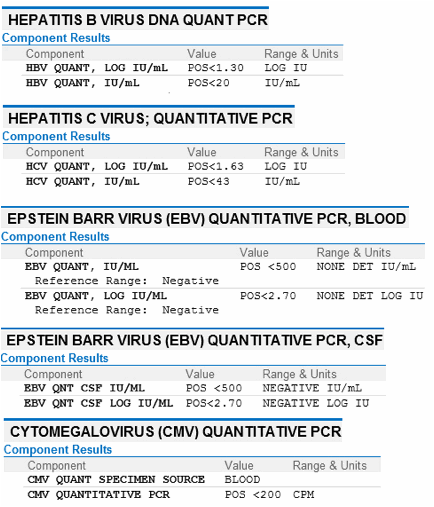
Hepatitis C Rna Pcr Qualitative Blood Test
The Hepatitis C RNA PCR Qualitative test is used to look for infections with the Hepatitis C virus. This test looks for the genetic material of the virus. Because viral genetic material may be detectable earlier than antibodies which develop in response to an infection, PCR testing can be used to screen for a recent exposure. This test is also useful as a confirmation for people who have had a positive result from a HCV Abs test. Results for this test are qualitative meaning they will come back as positive or negative.Hepatitis C is a virus spread through contact with infected blood. Nearly 80% of Hepatitis C infections develop into chronic Hepatitis. The number of people worldwide with chronic Hepatitis C infections is around 150 million. Chronic Hepatitis C infections can lead to serious health complication such as Cirrhosis and Liver Cancer. Many HCV infections display no symptoms. When symptoms do occur, some of the most common include:
Fever Grey feces Jaundice
Note: Result turn around times are an estimate and are not guaranteed. Our reference lab may need additional time due to weather, holidays, confirmation/repeat testing, or equipment maintenance.
Detection Period:
The Hepatitis C PCR test can typically detect the virus about 3 weeks from a suspected contact or exposure or anytime after. Some people may be detectable earlier.
Requirements:
Roche Taqman Hcv Assay
The Cobas TaqMan HCV test is a real-time nucleic acid amplification assay for quantitative detection of HCV RNA in human serum or plasma. Like the TaqMan HCV analyte-specific reagent , this assay was developed for use with the recently introduced Cobas TaqMan 48 analyzer . Amplification and detection were performed according to the manufacturer’s instructions for TaqMan HCV with a CTM 48 analyzer and Amlilink software, v. 3.0.1 . The lower limit of detection is 15 IU/ml, with 95% probability, using 1 ml of serum .
Requisitions And Kit Ordering
| < 1.50E+01 IU/mL | < 1.00E+03 IU/mL | HCV RNA detected below the lower limit of quantitation. Unable to quantify. or 1000 IU/mL .) |
| 1.50 E+01 to 1.00 E+08 IU/mL | 1.00 E+03 to 1.00 E+08 IU/mL | Viral load will be reported in IU/mL. |
| > 1.00E+08 IU/mL | > 1.00E+08 IU/mL | HCV RNA detected above the upper limit of quantitation. Unable to quantify. |
1Based on internal validation studies performed at PHO Laboratory, HCV RNA testing conducted on DBS is less sensitive than venous-collected samples. The lower limit of detection of HCV RNA using two DBS per test is approximately 1.6 to 2.0 logs higher than a concomitantly tested EDTA plasma or serum sample thus, DBS samples should NOT be used to rule out active HCV infection or to determine whether a patient on treatment has achieved an undetectable HCV RNA level.
No additional sample is usually required for HCV genotyping, provided there is sufficient volume. The first pre-treatment sample submitted for HCV RNA viral load testing will be used to automatically perform HCV genotyping if the HCV viral load is 500 IU/mL. Below this level, HCV genotyping cannot be performed.
Recommended Reading: How Can You Get Rid Of Hepatitis C
Extraction Of Nucleic Acids
Nucleic acids were extracted from 200 L of serum using a QIAamp MinElute Virus Spin kit according to the manufacturer’s suggested protocol. The concentration and quality of the extracted DNA and RNA were assessed by Nanovue spectrophotometry and by amplification of a fragment of the gene coding for -actin . The extracted DNA and RNA were stored at 80 °C until use.
Was Bedeuten Positive/erhhte Beziehungsweise Negative Hcv
Ein positiver Nachweis von HCV-RNA im Blut kann folgende Ursachen haben:
- akute Hepatitis C
- chronische Hepatitis C
Ein negativer HCV-RNA-Test schließt eine HCV-Infektion mit hoher Wahrscheinlichkeit aus. Im Anschluss an eine akute Hepatitis C muss der HCV-RNA-Nachweis über einen längeren Zeitraum negativ bleiben, bevor von einer Ausheilung gesprochen werden kann.Bei der Therapie der chronischen Hepatitis C bedeutet ein negatives HCV-RNA-Ergebnis, dass die Behandlung erfolgreich ist. Allerdings müssen hier regelmäßige Verlaufskontrollen durchgeführt werden.Mehr Informationen zum Thema
Don’t Miss: Does Hepatitis Have A Cure
What Is The Role Of Quantitative Hepatitis C Virus Rna Assays In The Diagnosis Of Hepatitis C Infection
Quantitative assays ascertain HCV RNA quantity in blood, using signal amplification or target amplification techniques . RT-PCR is more sensitive than bDNA testing. The HCV RNA level in blood helps predict the likelihood of a response to treatment, and the change in HCV RNA level can also be used to monitor the therapeutic response.
The same quantitative test should be used throughout therapy to avoid confusion, and results should be reported in international units to standardize data. The Versant HCV RNA Assay, version 3.0, is based on bDNA technology and has a dynamic range of 615-7,700,000 IU/mL. Another FDA-approved HCV quantitative test is the Aptima HCV Quant Dx Assay its limit of detection is 3.9 IU/mL in plasma and 3.4 IU/mL in serum.
The following are the best laboratory evidence of acute HCV infection:
- A positive HCV RNA test in the setting of a negative HCV antibody test
- A positive HCV antibody test after a prior negative HCV antibody test
It should be noted that impaired antibody production in immunosuppressed individuals may result in misleading information.
References
World Health Organization. Hepatitis C: fact sheet. Available at . Updated: October 2017 Accessed: January 23, 2018.
Frank C, Mohamed MK, Strickland GT, et al. The role of parenteral antischistosomal therapy in the spread of hepatitis C virus in Egypt. Lancet. 2000 Mar 11. 355:887-91. .
Hepatitis C Viral Load / Hcv Rna Quantitative Testing
Hepatitis C
The viral load of hepatitis C refers to the amount of virus present in the bloodstream. The quantitative HCV RNA tests measure the amount of hepatitis C virus in the blood. The result will be an exact number, such as “1,215,422 IU/L.” Many people refer to the quantitative measurement as the hepatitis C “viral load.”
Viral load tests are used to confirm active hepatitis C infection and are used during treatment to help determine response. If you have lower levels of virus in your blood when you start treatment, you may have a better chance of getting rid of the virus.
Recommended Reading: How Dangerous Is Hepatitis B
Optimization Of The Qpcr Assay
The qPCR assay was optimized following the MIQE guidelines. Primer and probe matrix experiments were conducted by selecting, for each gene, the primer concentration that resulted in the lowest cycle threshold and the highest Rn using a fixed amount of target template. Reactions with different concentrations of primers and probes were performed in total volumes of 25 L and 12.5 L, of TaqMan Universal Master Mix buffer sense primer and antisense primer concentrations of 50 nM, 300 nM, and 900 nM each probes at concentrations of 80 nM, 125 nM, 150 nM, and 250 nM and 1 L of DNA or cDNA. The reaction was performed with a 7500 Real-Time PCR System using universal conditions: 50 °C for 2 min, 95 °C for 10 min, 45 cycles of 95 °C for 15 s, and 60 °C for 1 min. For these reactions, an OptiQuant HBV/HCV Quantification Panel was used in the following concentrations expressed in international units/mL : HBV 7 or HCV 7 and HBV 2 or HCV 2 .
Other Things To Know:
- The viral load measurement does not tell us anything about the severity of a patient’s liver disease or the degree of fibrosis . For that information, the patient would need additional testing.
- It is not necessary to check the viral load repeatedly during treatment.
- If a quantitative HCV RNA result is reported as “< 15 IU/L,” this means that the quantitative test cannot measure the hepatitis C virus. It may mean that there is no detectable HCV RNA at all, but it may mean that the level of virus is just too low for the test to pick it up.
Also Check: Homeopathic Medicine For Hepatitis C
Abbott Realtime Hcv Assay
The RealTime HCV assay was performed at Abbott Laboratories according to the manufacturer’s specifications. Total nucleic acids were extracted from 0.5 ml serum or plasma by magnetic microparticle technology, using an Abbott m1000 automated sample preparation system. The internal control , derived from the hydroxypyruvate reductase gene of the pumpkin plant, Cucurbita pepo, was introduced as armored RNA into the sample lysis buffer. The IC was processed simultaneously with each sample. RNA was captured by magnetic microparticles, washed to remove unbound sample components, and eluted. A second step consisted of adding master mix to extract nucleic acid samples into 96-well optical reaction plates. Plates were sealed and placed on an Abbott m2000rt instrument for reverse transcription, PCR amplification, and detection/quantitation.
The HCV- and IC-specific probes are each labeled with a different fluorophore, thus allowing for simultaneous detection of both amplified products at each cycle. The amplification cycle at which a reactive level of fluorescent signal is detected by the m2000rt instrument is proportional to the log of the HCV RNA concentration present in the original sample. The homogeneous format and sealed PCR tray eliminate contamination by amplified products.
The Quantitative Hcv Rna Test Is Checked Before A Patient Starts Treatment
For each patient, the result can be described as either a “high” viral load, which is usually > 800,000 IU/L, or a “low” viral load, which is usually < 800,000 IU/L. It’s not uncommon to have a viral load in the millions. Today’s hepatitis C treatments are very effective with both high and low viral loads. An undetectable HCV viral load 10-12 weeks after hepatitis C is completed is associated with a cure.
You May Like: What Causes Hepatitis B And C
What The Qualitative Results Mean
The qualitative results indicate that HCV is present in your blood. The test result will be either detected or undetected.
Detected means that you do have the virus in your blood. Undetected means that you dont have the virus in your blood, or you have a tiny amount that cant be detected by this test.
The qualitative test results may still be positive even if your viral load has decreased drastically due to treatment.
Results From The Qualitative Test
Doctors use the qualitative HCV RNA PCR test to determine whether or not the hepatitis C virus is present in the blood.
If the virus is present, the test will be positive. If the test does not detect the virus, the result will be negative.
If the result is positive, a person will then need a quantitative HCV RNA PCR test. For this reason, many doctors now prefer to skip the first test and use the quantitative test straight away.
The quantitative test results show how much HCV is in the body. However, whether low or high, the viral load does not reflect levels of damage to the liver.
Other blood tests, ultrasounds, and, rarely, a liver biopsy will help a doctor determine overall liver health.
After using an HCV RNA PCR test to confirm the presence of HCV, doctors will work out which strain of the virus is active in the body. This helps a doctor plan the course of treatment.
The primary goal of treatment is to bring down the viral load in the body until it is entirely free of the virus. Doctors know this as a sustained virologic response .
SVR occurs when the virus is undetectable for 12 weeks or longer after treatment.
Achieving SVR is the best outcome of treatment, as it often means the person is free from hepatitis C, or that treatment has cured hepatitis C.
Doctors will also combine treatments with other tests that monitor for complications of HCV, including cirrhosis and liver cancer.
Recommended Reading: Can You Get Hepatitis From Sex
Preparation Prior To Transport
Label the specimen container with the patients full name, date of collection and one other unique identifier such as the patients date of birth or Health Card Number. Failure to provide this information may result in rejection or testing delay.Place specimen in a biohazard bag and seal. It is recommended to ship specimens for testing to PHO Laboratory immediately after collection or processing to avoid delays in testing. Whole blood that has not been centrifuged must be received at PHO Laboratory within 6 hours of collection, before 2:00 p.m. Monday – Friday. Serum stored at 2°C – 8°C must be shipped with ice packs within 6 days of separation. Frozen serum must be shipped on dry ice.Filter cards containing DBS specimens should be shipped to the PHO Laboratory in individual resealable bags containing a desiccant sachet. When the sample is appropriately prepared, it will be stable at room temperature for 30 days. Do not refrigerate samples. Do not ship on weekends.Shipping of specimens shall be done by TDG certified individuals in accordance with TDG regulations.
Who International Standard For Hcv And Synthetic Hcv Positive Control
The 5th WHO International Standard for HCV was purchased from National Institute for Biological Standards and Control and reconstituted in 1.1mL of nuclease-free water to 100,000 IU/mL . The HCV Positive Control prepared in-house is a non-infectious recombinant MS2 encapsidated construct containing the same 92-base target sequence derived from genotype 1a HCV and is amplifiable by the same set of HCV specific primers for the assay. The concentration of the HCV Positive Control was calibrated with reference to the 5th WHO International Standard for HCV.
Recommended Reading: Different Ways To Get Hepatitis C
What To Expect During Testing
A healthcare provider will take a blood sample for analysis.
Before the test, let them know if youre uncomfortable with certain needles or if youve ever passed out at the sight of blood. They can give you a snack to reduce your risk of fainting.
The needle may sting a little as it enters your skin, and you may have a bruise on the site of the draw for a few days.
Results are usually available within a few days or a few weeks at most.
The HCV RNA PCR test is conducted through a process called polymerase chain reaction . There are two approaches to this process: qualitative and quantitative.
What To Know About Hepatitis C Testing
The HCV RNA PCR test is a blood test that helps a doctor diagnose hepatitis C. The test measures the level of the hepatitis C virus in the bloodstream.
Hepatitis C is an infection that causes scarring in the liver and reduces function in this vital organ. Severe HCV can lead to liver failure. However, early diagnosis can reduce the risk of severe infection.
In this article, we look at how the test works and what the results mean.
The HCV RNA PCR test is a blood test. A lab technician looks for the genetic material of the HCV virus, or its ribonucleic acid . They use a process called a polymerase chain reaction .
The results of the HCV RNA PCR test help a doctor recommend different ways of reducing the viral load. The viral load indicates how many HCV viral particles are in the blood.
If a doctor suspects that a person has HCV, they will recommend this test early on in the diagnostic process, even if it is not the first test they carry out.
The test can detect the presence of the virus itself, rather than the antibodies that the body creates in response to the virus.
This means that a person does not have to wait until symptoms of the infection develop for a diagnosis.
It can take an average of 68 weeks for antibodies to become detectable after an HCV infection begins. However, a doctor can identify the virus itself after about 1-2 weeks by using PCR or another means of direct virus detection.
Doctors use the HCV RNA PCR in one of two ways:
You May Like: What Hepatitis Vaccines Are Available
Sera For Hcv Rna Analysis
A total of 177 serum samples were obtained from the 59 patients. Each of the specimens was divided into four aliquots and frozen to 80°C within 2 h of collection . Each aliquot was used for HCV RNA quantitation by the following assays: Cobas TaqMan HCV , with a detection limit of 15 IU/ml , and the Abbott RealTime HCV quantitative assay , with a detection limit of 10 IU/ml.
The HCV RNA viral load was measured at the following three time points: baseline and 4 and 12 weeks after the beginning of therapy. SVR was determined 6 months after the end of therapy with the Amplicor qualitative assay . Values for HCV RNA are reported in IU/ml. All assays were done at Alphabio Laboratory.
Viral Loads During Treatment
Checking your virus count before, during, and after treatment tells your doctor if and how well your drugs are working. A rising viral load doesnât always mean youâre getting sicker, and a drop in the virus count isnât a sign that youâre on your way to being cured.
Unlike with HIV, where lower viral counts usually mean longer, healthier life, HCV viral loads donât say much about how fast your hep C is progressing or how your disease might turn out. For that, your doctor will need to check your liver enzymes and your liver tissues and run other tests.
Usually, your hep C treatment will be the same no matter how high or low your viral load is. Your doctor will use your virus levels to monitor how you respond to the medication. The drugs youâre prescribed will depend less on your viral count than on your overall health, genetic makeup of your HCV, and other things.
Show Sources
Don’t Miss: What Type Of Hepatitis Can Be Cured
Hepatitis C Rna Quantitative
- Hepatitis C RNA, Quantitative
- Description
-
The Abbott real-time FDA approved HCV assay detects and quantifies Hepatitis C virus by real-time PCR amplification. The Abbott assay utilizes three master mixes, with each mix containing three primer/probe sets, with 1a and 1b subtyping in the NS5b region, and the remaining primer sets are in the 5UTR region. The RNA concentration detected with a probability of 95% or greater is 12 IU/mL .
For Tissue Biopsy, Bone Marrow, Swab, or Cellular fluid , see Hepatitis C Qualitative PCR, Tissue .
For HCV core sequencing, see HCV Genotype by Sequencing .
- Synonyms
- HCV RNA Quant, Hepatitis C by PCR, PCR-Hepatitis C, Viral Load-Hepatitis C
- Components