Identification Of Hbv Genotype Mixtures
DNA extracts were genotyped by multiplex PCR to identify the most predominant HBV genotypes in Mexico . These PCRs amplified different parts of the polymerase gene, a 370 bp fragment for genotype A, 147 bp for genotype D, and 279 bp for H, while a 584 bp fragment of the core gene was amplified for genotype G . The reaction was prepared with 2 L of DNA extract, 1 L of each genotype-specific primer set , 8.5 L of water, and 12.5 of 2× Hot StarTaq Master Mix . Thermal cycling conditions were as followed: preheating at 95°C, 15 min and 35 amplification cycles at 94°C for 1 min, 60°C for 1 min, 72°C for 2 min, and a final extension at 72°C for 10 m. DNA amplicons were run on a 2% TBE-agarose gel stained with SYBR Safe DNA gel and evaluated under UV light. The sizes of PCR products were estimated based on the migration pattern of a 50-bp DNA ladder . Quality control procedures included adding positive and negative controls in each PCR assay, and standard precautions were taken to avoid cross-contamination. The primer sequences designed for genotypes H and G were previously published , whereas primers for genotypes A and D were used as published by Kirschberg et al. . Mixtures of HBV genotypes were defined as finding two or more products in agarose gel, and the genotype was determined according to the bands size of the amplified DNA.
Facts About Hepatitis B Virus
- It is the commonest cause of viral hepatitis
- It can be contracted through blood contacts, unprotected sex and mother to child transmission.
- It is a cause of liver cancer
- Common drugs for hepatitis b infection are lamivudine, interferon, abacavir, entacavir, tenofovir and so on.
- About 23 million Nigerians are infected with Hepatitis B
- That hepatitis b is curable is still controversial
- The common tests for hepatitis b are HbsAg, liver function test, HBV genotype, Hbeag, anti-Hbe and hepatitis b viral load.
- Treatment for hepatitis b is expensive.
- It is best prevented by vaccination.
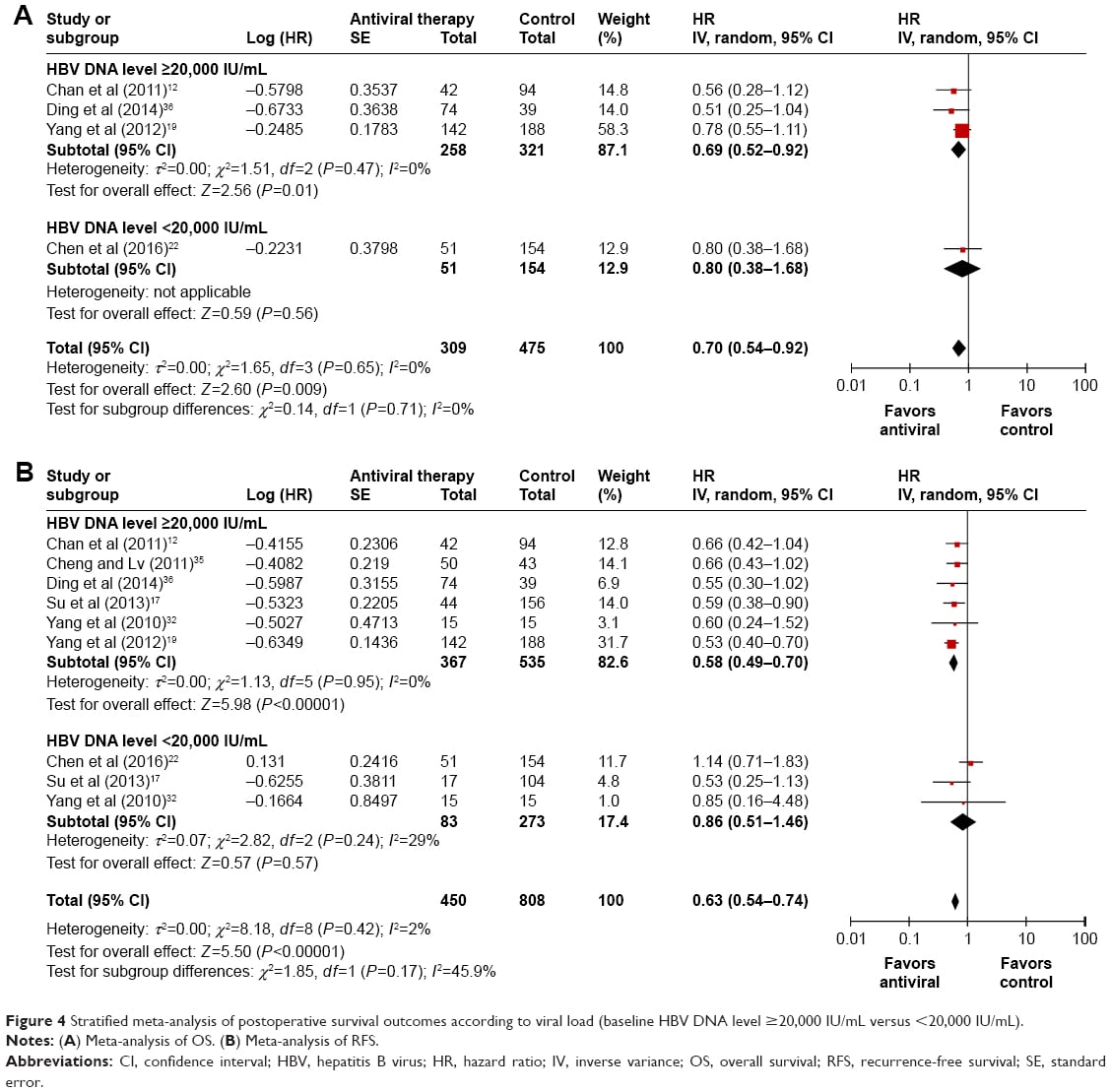
Viral Load And Treatment: Unfavorable Arguments
Presently, the viral load cannot be considered as the only treatment criterion. HBV DNA persists even in persons who have serological recovery from acute HBV infection. Patients with low HBV-DNA levels, between 300 and 104 copies/mL, have, although a very low one, a risk of progression to cirrhosis and HCC. The progression in CHB infection is a multi-factorial process including interaction between host and environmental factors.
You May Like: Hepatitis B Surface Antibody Ql Reactive Means
Understanding Your Test Results
Understanding your hepatitis B blood tests can be confusing. It is important to talk to your health care provider so you understand your test results and your hepatitis B status. Are you infected? Protected? Or at risk? The Hepatitis B Panel of blood tests includes 3 tests and all three results must be known in order to confirm your status.
Below is a chart with the most common explanation of the test results, but unusual test results can occur. Please note that this chart is not intended as medical advice, so be sure to talk to your health care provider for a full explanation and obtain a printed copy of your test results. In some cases, a person could be referred to a liver specialist for further evaluation.
More Detailed Information About Hepatitis B Blood Tests
An acute hepatitis B infection follows a relatively long incubation period – from 60 to 150 days with an average of 90 days. It can take up to six months, however, for a person to get rid of the hepatitis B virus. And it can take up to six months for a hepatitis B blood test to show whether as person has recovered from an acute infection or has become chronically infected .
The following graphic from the U.S. Centers for Disease Control and Prevention represents the typical course of an acute hepatitis B infection from first exposure to recovery.
According to the CDC, a hepatitis B blood test result varies depending on whether the infection is a new acute infection or a chronic infection.
Interpreting Hepatitis B Laboratory Results
Many jurisdictions have regulations requiring laboratories to report all positive HBsAg, HBV DNA, and anti-HBc IgM laboratory results to the HD while a subset might also routinely receive positive total anti-HBc and anti-HBs results.
Additionally, some HDs might receive negative hepatitis B laboratory results, which are useful for determining false-positive results and monitoring patients through their infection and recovery. Table 3-1 shows how to interpret the combinations of laboratory results frequently available in hepatitis B test panels, following the biomarker changes over the course of disease as shown in Figure 3-1.
Table 3-1. Interpretation of hepatitis B laboratory results
| HBsAg |
|---|
- Concurrent ALT and total bilirubin result
- Other hepatitis serological results
- Negative HBsAg and/or negative/undetectable HBV DNA results
Total anti-HBc is detectable, on average, approximately 5 weeks post-HBV exposure, remains detectable indefinitely following exposure, and indicates past or current infection. In the presence of total anti-HBc, a positive HBsAg, HBeAg, or anti-HBc IgM result is a more reliable indication of recent or current infection. Jurisdictions that receive total anti-HBc laboratory results can use these results to clarify a persons HBV infection status.
Don’t Miss: What Is A Hepatic Diet For Dogs
Where Can You Do Hepatitis B Viral Load Test
It can be done in many big labs in Nigeria especially in Lagos, Abuja, Port Harcourt, Ibadan, Benin city, Delta, Kaduna, Kano and so on. In Lagos, you can do it in Nigerian Institute of Medical Research in Yaba. Their results are reliable. Labs like Pathcare , Union Diagnostics, and Raytouch does Hepatitis b DNA test. Teaching hospitals and Federal Medical Centres also do the test.
Relationship Between Hbv Mixtures And Clinical Characteristics
We compared different clinical parameters as dependent variables relative to the number of HBV genotypes , finding a positive correlation between increasing age and mixtures . Interestingly, the median age of patients with two HBV genotypes was 37.0 years, while the age-associated with triple-mixed cases was 42.0 years . Moreover, platelet levels were negatively correlated with the increase in the number of HBV genotypes . HBV viral load was higher in triple-mixtures than dual-mixtures . Regarding liver damage, the degree of fibrosis was similar between single and dual genotype mixtures , while the liver fibrosis grade was significantly higher in triple-mixtures than dual-mixtures . Based on APRI, patients with HIV and three HBV genotypes had a high risk of presenting significant liver fibrosis compared with dual-mixtures or single genotype infection .
Figure 3. Shows a significant relationship between increasing age and the presence of a mixture of HBV genotypes. , Platelet levels decreased while increasing the number of HBV genotypes. Shows viral load levels by type of mixes. Shows the comparison of the degree of fibrosis in patients with one, two, and three HBV genotypes, evaluated through the APRI, FIB-4, and LSM, respectively. The boxes middle lines represent the median, while the outer lines represent 25 and 75 quartiles. Black dots indicate each patient studied. LSM, liver stiffness measurement.
You May Like: What Is Autoimmune Hepatitis C
Value Of Biopsy: Fibrosis And Necro
Patients with moderate/severe inflammation or bridging fibrosis/cirrhosis must be treated. The degree of fibrosis or inflammation upon liver biopsy cannot be predicted for HBV-DNA levels > 10000 copies/mL. ALT is an imperfect marker for liver disease . Traditional and current guidelines recommend liver biopsy for patients who meet the criteria for chronic hepatitis . These patients must be treated if we follow the guidelines, and we believe that liver biopsy may be unnecessary.
Liver biopsy is more important for patients who do not meet the current criteria for treatment but have serum HBV-DNA levels of 104 to 105 copies/mL and/or ALT/AST levels that are normal or mildly elevated . The presence of significant inflammation or bridging fibrosis/cirrhosis is an indication for treatment. In a subgroup of these patients, hepatic elastography can avoid the need to carry out a liver biopsy for detection of significant fibrosis. This is a novel non-invasive method to assess hepatic fibrosis in patients with a chronic disease, by measuring liver stiffness. Its failure rate is about 5% of cases, mainly in obese patients. Elastography has a high positive predictive value for diagnosing significant fibrosis in Asian CHB patients. This method does not have a defined role in HBeAg-negative patients because histological inflammation during reactivation may affect the results.
Qualitative Detection Of Hbv Dna
The yield of DNA extracts was tested by nested PCR. The amplification reaction was prepared with 5 L of DNA, 1 L of each primer , 0.5 L of dNTPs mix , 1 L of MgCl2 , 2.5 L of PCR buffer , 13.8 L of water and 0.2 L of Taq DNA polymerase in a final volume of 25 L. The first round amplified a 418-bp fragment of the surface gene under the following conditions: 94°C for 3 min followed by 40 cycles of 94°C for 30 s, 55°C for 30 s, 72°C for 30 s, with a final extension step of 72°C for 5 min. The second round amplified a 232-bp product under the same conditions.
Also Check: How Do You Contract Hepatitis D
Cases And Clusters Of Potential Public Health Importance
Jurisdictions should review and analyze hepatitis B data regularly to identify cases and clusters of hepatitis B that merit further investigation. When resources are limited, these should be prioritized for investigation based on the degree of public health importance. The following are examples of high priority cases and clusters:
- People of childbearing age who are or have the potential to become pregnant, indicating the potential for perinatal transmission
- Children 24 months of age to detect perinatal transmission
- People in age and demographic groups for whom infection may be acute due to recent transmission, including those
- 70 years of age
Hbv Serological Markers And Viral Load
All serum samples were tested for hepatitis B surface antigen using an enzyme-linked immunosorbent assay according to the manufacturers instructions. In samples positive to HBsAg, HBV viral load was quantified by real-time polymerase chain reaction , which has a detection limit between 20 and 170 million IU/mL . Levels 2,000 IU/mL was considered as high viral load and < 2,000 IU/mL as low viral load.
Recommended Reading: Who Should Get Tested For Hepatitis C
My Hepatitis B Viral Load Is Low Am I Still Infected With Hepatitis Delta
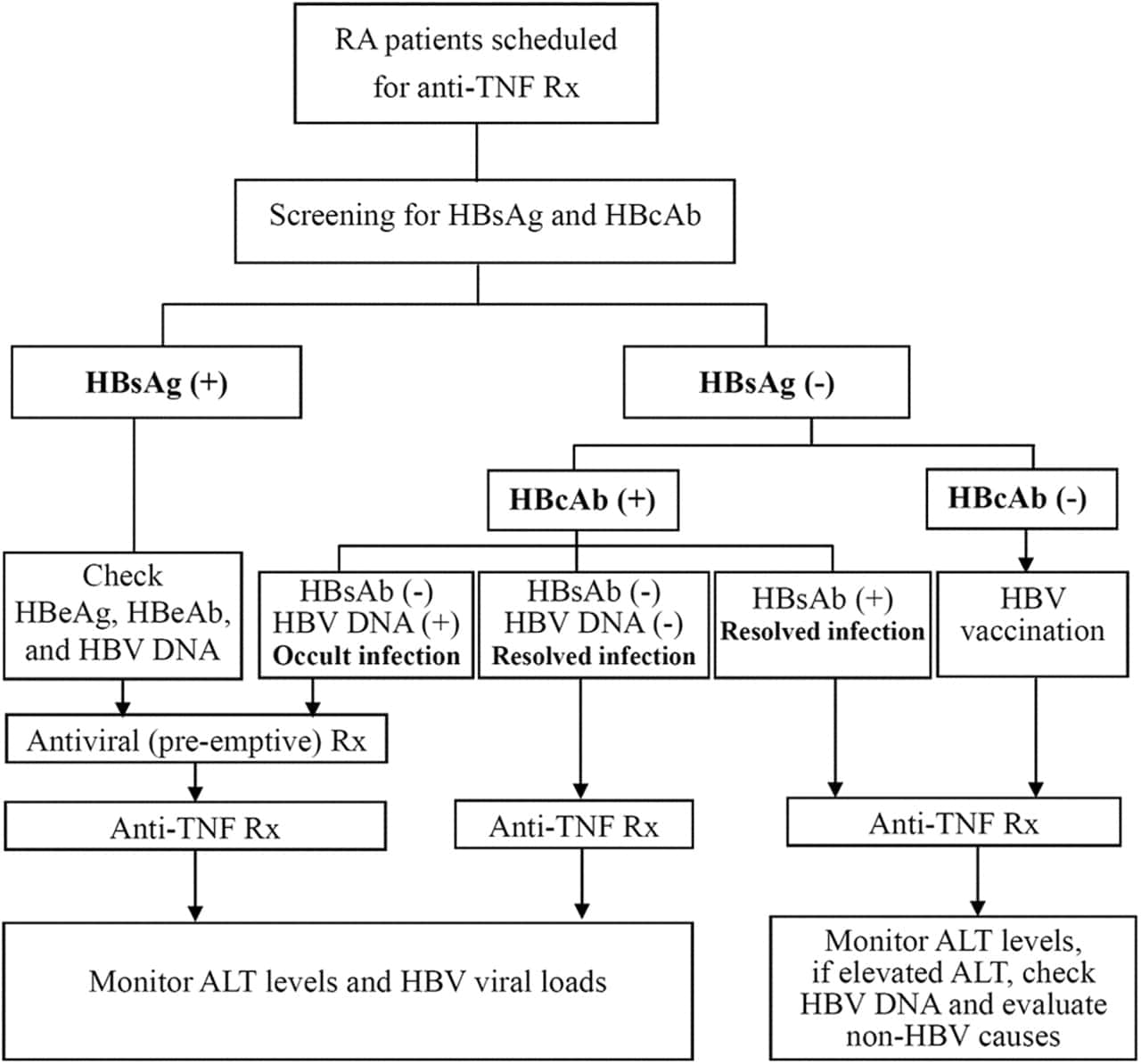
For people who have been diagnosed with chronic hepatitis B and delta coinfection, a low or undetectable hepatitis B viral load does not usually indicate that theyve cleared both infections. This is because, in cases of coinfection, hepatitis delta usually becomes the dominant virus, and suppresses hepatitis B, slowing or even stopping its replication entirely. If someone is still positive for the hepatitis B surface antigen , the hepatitis delta virus can still replicate and cause potential liver damage 1. For this reason, the test to measure hepatitis delta activity, the HDV RNA test, is important in disease monitoring and management 2,3. Available since 2013, the HDV RNA test can be acquired internationally through the Centers for Disease Control and Prevention , and from several labs in the US.
For those suspected of having acute hepatitis B and delta coinfection, HBsAg testing should follow 6 months after initial diagnosis. If HBsAg is negative , both infections are likely to have cleared. Its important to remember that people who contract hepatitis B and delta during one exposure are likely to clear both viruses. If HBsAg is positive after 6 months, both infections are likely chronic . Those who are known to have a chronic hepatitis B infection and then become infected with hepatitis delta later on, they are likely to develop chronic coinfections.
Both hepatitis B and delta are prevented with the safe and effective hepatitis B vaccine series.
References:
Core Antigen And Antibody
Hepatitis B core antigen
The hepatitis B core antigen is part of the nucleocapsid within HBV it is not routinely measured in clinical practice however, the body produces a corresponding antibody to this antigen called hepatitis B core antibody , which is clinically relevant.
Antibody to Hepatitis core antigen :
Anti-HBc is the antibody produced by the host in response to HBcAg depending on which immunoglobulin type is present indicates the time frame since infection. The presence of anti-HBc IgM indicates recent infection within the last six months.1 Over time, IgM is gradually replaced by anti-HBc IgG therefore, anti-HBc is seen in patients with resolved infection and those with chronic infection.1 This is shown in Figure 2, as anti-HBc IgM acutely rises and falls, whilst total anti-HBc rises and plateaus as IgG production predominates.6
Figure 2
Read Also: Hepatitis A What Is It
Study Design And Socio
This cross-sectional study included 228 patients with a serological or molecular diagnosis of HIV. All patients were attended at the Department of Molecular Biology in Medicine and the HIV Clinic of the Civil Hospital of Guadalajara Fray Antonio Alcalde from January 2014 to December 2016 . Written informed consent was obtained from all participants. Demographic and clinical information, such as gender, age, HIV viral load, and the combination antiretroviral therapy received during recruitment, were obtained from the patients medical records. The levels of liver enzymes, platelets, albumin, CD4 cells, and CD8 cells were obtained from our hospitals general laboratory. Also, hepatitis C virus infection was assessed by the presence of anti-HCV antibodies . The methodological summary of this study is shown in Figure 1.
Figure 1. Workflow used to detect mixtures of HBV genotypes in patients with HIV. First, serum samples were collected from all patients. These samples were analyzed by ELISA to evaluate the presence of HBsAg. Next, in the positive samples from the previous step, the viral load levels were measured by real-time PCR. A nested PCR was used to confirm HBV infection in the samples with detectable viral load. After that, a multiplex PCR was used to detect the predominant HBV genotypes in Mexico. Finally, the products were sequenced and genotyped by Sangers method and phylogenetic analysis, respectively.
Hbv Dna Hbv Genotype And Hbv Drug Resistance Assays
Specimen: Serum or plasma
Container: Red-top tube, yellow-top tube , gel-barrier tube, plasma preparation tube, or lavender tube
Collection method: Routine venipuncture
The specimen should be transfused to separate plasma/serum from cells within 6 hours and kept frozen when testing cannot be done promptly.
The tests use PCR amplification, DNA probe hybridization, and sequencing method.
Read Also: What Test For Hepatitis B
General Characteristics Of The Study Population
A total of 228 patients with HIV were analyzed most of them were men between 32 and 46 years of age . At the time of this study, 82.0% of the participants were on antiretroviral therapy, with a median HIV viral load of 40.0 IU/mL . Among the 187 treated patients, the medical records revealed that cART was tailored individually in which none were monotherapy. All received at least one drug that is known to affect the HBVs life cycle, such as tenofovir, lamivudine, or emtricitabine. However, the most frequent combinations were tenofovir + emtricitabine + efavirenz in 39.6% , followed by tenofovir + emtricitabine + lopinavir + ritonavir in 11.2% and 49.2% received diverse unique combinations with these and other antiviral drugs. Also, 62 individuals out of 132 had evidence of hepatitis C virus infection . Overall, liver injury was detected in 24.6% , of which 18.3% had significant liver fibrosis, 6.9% had advanced fibrosis, and with liver stiffness, 22.5% had advanced liver fibrosis .
Table 1. Demographic data of patients with HIV and HIV/HBV.
Table 2. Baseline clinical characteristics of patients with HIV and HIV/HBV.
Table 3. Baseline non-invasive markers of liver damage in HIV and HBV/HIV patients.
What A Hepatitis B Viral Load Test Can Tell You About Your Infection
The hepatitis B virus DNA quantification, also known as the viral load test, is a blood test that measures the amount of hepatitis B virus DNA in the blood of chronically infected patients. The blood is tested using a Polymerase Chain Reaction technique. By itself, the viral load shouldnt tell you all that much. The information gained from an HBV viral load test should only be considered in relation to other information like your e-antigen status, liver enzymes test results, and inflammation levels. The viral load is usually measured in international units per milliliter , but in the past, it was measured in copies per milliliter.
Viral load varies over time, depending on the phase of your hepatitis B infection. Its critical to have your viral load regularly monitored to fully understand what stage of the virus you might be experiencing. The phase you are in is determined by an array of lab results, which includes viral load.
Chronic hepatitis B infection is characterized by four phases. Not everyone will experience all four phases, and the lengths of the phases vary among people. Many people living with hepatitis B will transition between these phases.
Reactivation Phase: People who are HbeAg negative may experience reactivation. ALT loads are elevated, HBV viral loads are above 2,000 IU/mL, and liver inflammation and fibrosis levels are moderate-to-severe.
Don’t Miss: What Is Hepatitis C Screening Test