Target Prediction And Dual
The predicted miR-148a target genes were retrieved from the TargetScan , Miranda and miRTarBase database. Gas1, a glycosylphosphatidyl-inositol-linked membrane glycoprotein, was one of the potential candidates, that caught our attention because it encodes a Hedgehog surface binding receptor and facilitates Hh signaling and because of the putative miR-148a binding sequences in the 3-UTRs of the Gas1 mRNA.
To assess whether Gas1 was a direct target of miR-148a, a dual-luciferase® reporter assay was performed. Briefly, 293T cells were plated in a 24-well plate and co-transfected with 20 nM of miRNA control or miR-148a, 5 ng of pRL-TK using Lipofectamine 2000 and 10 ng of firefly luciferase reporter that containing the wild-type or mutant-type Gas1 3-UTRs. The pRL-TK vector was co-transfected as an internal control to correct for the differences in both transfection and harvest efficiency. Transfections were performed in duplicate and repeated in three independent experiments, and the results were normalized to pRL-TK luciferase activity. Luciferase activities were analyzed with a Dual-Luciferase Reporter Assay System on a Microplate Fluorescence Reader 48 h after transfection.
General Aspects Of Immortal Hsc
Primary HSC provide characteristics very close to that observed in vivo, but have some significant disadvantages. First, there may be considerable variation in the cellular features of cells prepared in different laboratories or from different operators. In addition, there are a number of plausible reports describing the occurrence of heterogeneity within HSC cultures. Secondly, cells originating from one preparation have only limited utility because of their limited life span. Thirdly, sub-cultivation of primary HSC can be achieved only for a limited number of passages associated with modified characteristics, apoptosis, altered cytokine susceptibility and variations in gene expression . Fourthly, experiments requiring large numbers of cells or long-term growth are rarely possible. Moreover, there is considerable pressure on scientists to reduce or even eliminate the use of animals in research.
To overcome these limitations, many investigators have transferred established concepts to develop permanent HSC cell lines. Presently, immortal HSC cell lines were derived from primary HSC that were transformed with the simian virus 40 large T-antigen which has pleiotropic effects on cell division achieving this by binding to the transcription factor E2, p53âor pRB, manipulated by ectopic expression of telomerase reverse transcriptase activity, isolated from experimentally diseased livers, subjected to UV light or alternatively were spontaneously immortalized during culturing.
Protein Extraction And Western Blotting
Proteins were extracted 48 h after transfection using 100 l of 1 × RIPA buffer . Protein concentrations were measured using BCA protein assay kit . Cell extracts were separated via 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and then transferred onto polyvinylidene difluoride membranes . The membranes were blocked with 5% skim milk in TBS/0.5% Tween-20 for 1 h at room temperature. Then, the membranes were incubated with specific primary antibodies overnight at 4°C with gentle shaking. After three washes with PBS-Tween-20 , appropriate secondary horseradish peroxidase -conjugated antibodies were used at 1:2,000, and the membranes were incubated for 1 h at room temperature. Goat anti-rabbit monoclonal antibodies for GAPDH were used for normalization. The results were visualized using an enhanced chemiluminescence detection system . All western blot results were scanned and semi-quantized using Image Pro-Plus 6.0 software .
Read Also: Dental Management Of Hepatitis Patient Ppt
Small Interfering Rna Transfection
To examine the influence of CHOP expression in LX2 cells treated with GGA, we performed experiments using siRNA transfection. RNAi was obtained from Dharmacon and transfected to LX2 cells according to the manufacturers protocol. Cells were used for experiments 24 h after transfection. To confirm knockdown of CHOP, RT-PCR was performed as described above.
Western Blotting And Immunoprecipitation
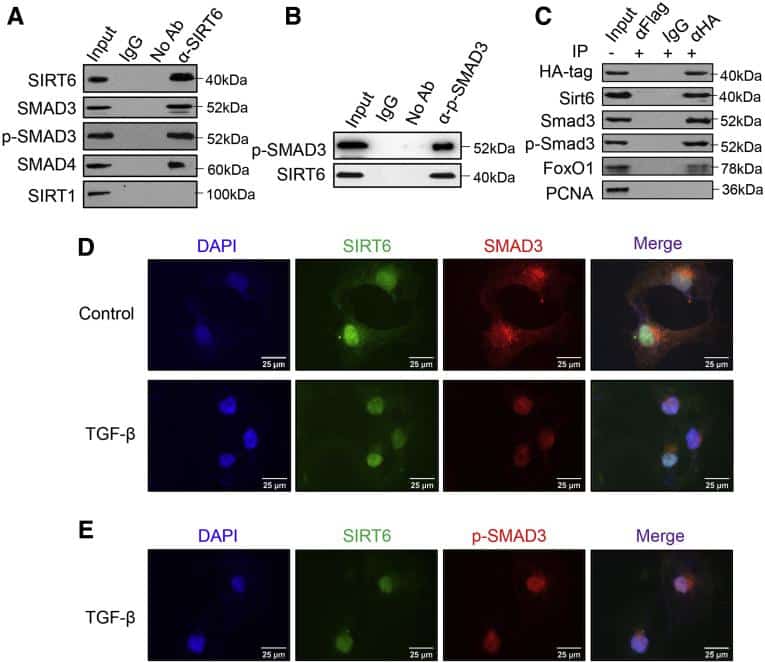
Cells lysates were extracted with radioimmunoprecipitation assay buffer containing 0.1% sodium dodecyl sulfate , 1% NP-40 and 0.5% sodium deoxycholate in phosphate-buffered saline . The samples were then mixed with loading dye before adding into a 10% SDS-PAGE polyacrylamide gel . Gel electrophoresis was run at 130V for 1.5 h. Subsequently, a wet transfer sandwich method was used to transfer the proteins from the gel onto PVDF membrane at 4°C with 100V for 2 h. The membrane was washed with Tris-buffered saline containing 0.1% Tween , blocked with 5% bovine serum albumin and incubated with primary antibody containing 2% BSA at 4°C overnight. The membrane was washed thrice with TBST, incubated with secondary antibody at room temperature for 1 h and exposed with Western Lightning Plus-ECL reagent using G:Box Gel imaging system . Original images of Western blot analyses are supplied in Supplementary Figure S6.
For immunoprecipitation of SMAD proteins, primary antibody was added into the cell lysates and incubated at 4°C overnight. Subsequently, 30 l of protein G Sepharose Fast Flow Beads were added and the samples were further incubated at 4°C for 4 h. The beads were centrifuged at 3,000 rpm for 1 min, washed with cold RIPA buffer for three times and boiled with RIPA buffer containing SDS and dithiothreitol at 95°C for 10 min. Finally, the beads were further centrifuged to collect the supernatant for Western blot analyses.
Read Also: How Can You Tell If You Have Hepatitis B
Propidium Iodide Staining And Cell Cycle Analysis
Cells were seeded at a density of 20,000 cells per cm2 onto a 6-well plate overnight before the addition of TGF- and MgIG. After 24, 48, and 72 h treatment, both the adhered live cells and floating dead cells were harvested, washed with cold PBS and mixed with 300 l of PBS containing 10 g/ml PI stain. The percentage of dead cells was then determined with Beckman Coulter CyAn ADP flow cytometer . Three biological replicates were performed and the average percentage of dead cells was plotted against treatment time. For cell cycle analysis, the cells were further permeabilized with 70% ethanol at 4°C for 1 h. Thereafter, the cells were sorted up to 20,000 events with FC 500 flow cytometer and assessed based on the DNA quantity. Nuclei number was then plotted against the measured DNA fluorescence. Non-treated cells were used as the negative control to determine the percentage of cells in different cell cycle phases.
Nadph Oxidase Inhibition And Farnesoid X Receptor Activation Rescue Phh Functions In The Presence Of Activated Hscs
In MPTCs , we observed an increase in the gene expression of NFE2L2 , a transcription factor involved in oxidative stress signaling . Reactive oxygen species generated via NOX1 and NOX4 enzymes can mediate fibrogenic pathways in HSCs and hepatocytes. We did not detect the expression of either NOX1 or NOX4 in MPCC controls, but both were detected in pure activated HSC cultures with NOX4 expression being higher than NOX1 . However, only NOX4 gene expression was detected in MPTCs, likely due to the contribution from the activated HSCs at the physiological seeding ratio. Since the inhibition of NOX1& 4 with the small molecule GKT137831 has shown anti-fibrotic effects in mouse models, we hypothesized that incubating MPTCs with GKT137831 could alleviate the hepatic dysfunctions observed due to the presence of activated HSCs. Furthermore, FXR is a major regulator of hepatocyte metabolism and bile production/transport that is downregulated in NASH patients. Activation of FXR via obeticholic acid has shown positive effects in ameliorating lipid accumulation and fibrosis in patients with NASH. Since we observed a strong downregulation of NR1H4 gene expression in PHHs, we hypothesized that treatment of MPTCs with OCA could alleviate hepatic dysfunctions. Finally, we tested the simultaneous effects of both drugs on MPTCs.
Don’t Miss: Va Disability Rating For Hepatitis C
Human Hepatic Stellate Cell Lines
There are obvious disadvantages in obtaining and usage of primary HSCs, particularly primary human HSCs, such as the heterogeneity of isolated cell populations and cellular characteristics, limited supply, considerable variations of cell preparation in different laboratories, as well as the isolation equipment and techniques requirements. Immortalized HSC lines were established and have been used in a wide range of research. These immortalized cell lines provide unlimited resource supply, homogeneity, and are suitable for genetic manipulation studies. They recapitulate many activated HSC features, and can serve as a useful tool for mechanistic investigation of HSC function in hepatic fibrosis and liver pathophysiological processes. The immortalized HSC lines currently in use have been generated from primary HSC through spontaneous immortalization during long-term culture, or by transformation with the simian virus 40 large T-antigen , or ectopic expression of human telomerase reverse transcriptase . Notably, none of the published cell lines are reported to be tumorigenic. Considering these cells are genetically modified, careful evaluation of the reported studies is always warranted .
Table 2 Characteristics of human hepatic stellate cell lines
What Gets Stored In A Cookie
This site stores nothing other than an automatically generated session ID in the cookie no other information is captured.
In general, only the information that you provide, or the choices you make while visiting a web site, can be stored in a cookie. For example, the site cannot determine your email name unless you choose to type it. Allowing a website to create a cookie does not give that or any other site access to the rest of your computer, and only the site that created the cookie can read it.
You May Like: The Early Signs Of Hepatitis C
Conventional And Molecular Karyotyping
To characterize LX-2 genetically, we first performed chromosome G-banding analysis using different LX-2 cell cultures. This analysis revealed that LX-2 cells have several numerical and structural chromosomal aberrations . The karyotype as assessed by this methodology was described as: 5578, deradd, Y , del , +del , der , +der , del , +delx2 , +deradd , +der?t , deradd , +deradd , +dert , der?t?del , deradd , +deraddx2 , +6 , del , +del , der , +der ,i , del , +del , 13 , 14 , +15 , +15 , dert , +dert , +der , +19 , +20 , +20 , +20 , +21 , +813mar , respectively.
Spectral karyogram of the hepatic stellate cell line LX-2.
Shown is a representative image of LX-2 cells hybridized with a 24-color SKY probe . The chromosomes were counterstained with DAPI . The spectral images of hybridized metaphases were acquired using a cytogenetic workstation and grouped with the SkyView software . Based on this analysis, the karyotype of this LX-2 metaphase is determined to: 73< 3n+> , XXXYY, ish +dertt, dert, del, +dert, derttdel, 5, +del, dert, dert, dert, del, +dertt, dert, +dertdel, derttdel, +derdeldel, dert, del, derdeldelx2, dert, 13, 14×2,+i, +dert, dert, del, 19, 20, 21, dert, del, del, del. Please note that the derivative that contains parts of chromosomes 1, 18 and 7 were also seen in this analysis.
Cellular Crosstalk Between Activated Hscs And Cellular Components Of The Hepatic Tme
The hepatic TME consists of various immune cells to create an immunosuppressed environment in order to maintain HCC tumor growth . Activated HSCs contribute to this immunosuppressed environment by secreting cytokines which induce MDSC expansion . In an orthotopic liver tumor mouse model, activated HSCs significantly increased regulatory T cell and MDSC expression to benefit HCC growth in the spleen, bone marrow, and tumor tissues . Furthermore, activated HSCs secrete angiogenic growth factors to form new vasculature within the TME . These functions of activated HSCs create a link to the circulatory system for supplying nutrients to the tumor. Immune cells may also regulate activation of HSCs in vitro, demonstrated by Interleukin 20 activation of HSCs, resulting in upregulation of TGFB1 and type I collagen, and increased proliferation and migration of activated HSCs . The same study further indicated that these fibrogenic phenotypes could be attenuated with an anti-IL-20 receptor monoclonal antibody, proposing IL-20 as a significant activator of HSCs and fibrogenesis. Taken together, activated HSCs may have an important role in promoting an immunosuppressed and angiogenic hepatic TME to support aggressive HCC cell growth.
Read Also: Antiviral Medication For Hepatitis B
Establishment And Characterization Of An Immortalized Human Hepatic Stellate Cell Line For Applications In Co
XiaoPing Pan1,2, Yini Wang1,2, XiaoPeng Yu1,2, JianZhou Li1,2, Ning Zhou1,2, WeiBo Du1,2, YanHong Zhang1,2, HongCui Cao1,2, DanHua Zhu1,2, Yu Chen1,2, LanJuan Li1,2
1. State Key Laboratory for the Diagnosis and Treatment of Infectious Diseases, First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, 310003, China.2. Collaborative Innovation Center for the Diagnosis and Treatment of Infectious Diseases, Hangzhou, China.
Corresponding author: LanJuan Li, M.D. Collaborative Innovation Center for the Diagnosis and Treatment of Infectious Diseases, State Key Laboratory for the Diagnosis and Treatment of Infectious Disease, First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou, 310003, China. Tel: 86-571-87236759 Fax: 86-571 87236759 Email: ljliedu.cn.More
Citation:Int J Med Sci
Gga Decreased Density Of Hscs
We monitored LX2 cells treated with GGA using phase-contrast microscopy and observed a concentration-dependent decrease in cell density . Similarly, GGA reduced the density of HHSteCs in a concentration-dependent manner. In contrast, HepG2 cells showed no significant morphological or density changes with GGA treatment.
Fig. 1
Geranylgeranylacetone decreased LX2 and HHSteC density in a concentration-dependent manner. After treatment with GGA for 24 h, three different cell lines, LX2 cells, HHSteCs and HepG2 cells, were observed by phase-contrast microscopy. There were no significant morphological changes or cell density decrease in HepG2 cells
Recommended Reading: Can Drinking Cause Hepatitis C
Synergistic Cytotoxicity Of Arsenic Trioxide In Combination With Fluorouracil In Hepg2 Cells
To reduce variability and facilitate multiple cell line and drug combinations, dose response and drug combination experiments were carried out in 96-well plate format using an automated, high throughput system. First, to determine the degree of cytotoxicity of 5-FU alone on the LX2 and HepG2 cells, both cell lines were treated with increasing concentrations of 5-FU for 24 and 72hr. Neither cell type experienced significant cytotoxicity following 24hr of treatment, but cytotoxicity was observed in HepG2 cells treated with 5-FU for 72hr . In contrast, LX2 cells remain resistant to 5-FU even with high drug concentrations and prolonged exposure .
Figure 3
5-FU inhibits HepG2 and LX2 Cell Proliferation at 72hrs. HepG2 and LX2 cells were treated with 5-FU at the indicated concentrations for 24 and 72hrs and cell proliferation was assessed by MTT assay. For each concentration, percent inhibition values were calculated and data normalized to vehicle control. IC50 values shown in the corresponding tables were determined by non-linear regression in GraphPad Prism .
Figure 4
Hepatocyte Growth Factor And Transforming Growth Factor
To measure HGF and TGF-1 secretion in the HSC-Li cells, we inoculated 1.0 Ã 106 of HSC-Li cells into 90 mm dishes. After 24 h of culture, the cells were cultured with DMEM media supplemented with low amounts of FBS for eleven days. The culture supernatant of the HSC-Li cells was collected daily and assayed using human HGF and TGF-1 ELISA kit according to the manufacturerâs manual.
You May Like: Hepatitis B Vaccine Schedule For Adults
Don’t Miss: Can Hepatitis C Spread Through Saliva
Culture And Growth Characteristics
Generally, primary HSC are maintained under standard conditions in medium containing 10% FCS. Under these conditions, they have an overall low doubling time of several days and can be passaged only two or three times before going through a crisis, entering senescence and ending in an enlarged and flattened phenotype. The underlying mechanisms of this replicative senescence are most likely inactivation of proliferation-promoting genes and/or activation of anti-proliferative genes or the switch from a fibrogenic to an inflammatory phenotype leading to apoptosis . Established HSC lines, however, grow faster with typical duplication times between 24 and 72 hrs and can be continuously passaged demonstrating that these cells have lost many characteristics of the primary cultures from which they are derived. In several lines, these changes of growth characteristic are accompanied by confounding genetic events introduced by an aneuploid chromosome complement that is characterized by additions or deletions of whole chromosomes from the expected balanced diploid number of chromosomes .
Combination Treatment With Arsenic Trioxide And Sorafenib Shows A Trend Towards Increased Cytotoxicity And Attenuates P
The induction of a delayed and sustained survival signal as seen by pERK and pJNK activation in HepG2 cells may potentially contribute to an ATO resistance mechanism. Based on observations in other systems that combination treatment with ERK or JNK inhibitors restores chemosensitivity, we hypothesized that treatment with ATO and the multikinase inhibitor, sorafenib, would be more efficacious than either agent alone. In order to evaluate the effect of sorafenib on cellular proliferation, HepG2 and LX2 cells were treated with increasing concentrations of drug for 24 to 96hr. Sorafenib has significant cytotoxic effects on both HepG2 and LX2 cells in a dose and time dependent fashion . To assess synergism of the two agents, HepG2 and LX2 cells were treated with 2.5 M ATO in the presence of increasing concentrations of sorafenib for 24hr . At 24hr there was no significant difference in IC50 values for the combination in comparison to sorafenib alone in both the HepG2 and LX2 cells. However at the later time points of 48 and 72hr, the percent of proliferating HepG2 cells was less for 20 M sorafenib+2.5 M ATO than for 20 M sorafenib alone . The increased cytotoxicity was accompanied by a decrease in phospho-MAPK levels in HepG2 cells treated with ATO and sorafenib in comparison to ATO alone . ATO treatment of LX2 cells results in potent cytotoxicity and the addition of sorafenib resulted in no further additive effect.
Figure 7
Also Check: Hepatitis C At Home Test
Analysis Of Retinoid Biology
Retinol uptake and processing by LX-1 and LX-2 cells
Following plating and overnight incubation, cells were treated with two different concentrations of retinol for 24 hours in the dark. For this purpose, a concentrated stock solution of retinol was diluted in a small volume of ethanol , added to the media with 300 M palmitic acid to aid esterification, and then vigorously mixed. Following incubation, media from triplicate cultures were removed and cells scraped into 2 ml of ice cold PBS. Samples were then stored at 20°C prior to analysis. Control cells were cultured in medium with ethanol vehicle.
HPLC analyses of retinol and retinyl esters
Retinol and retinyl esters were analysed by reversed phase high pressure liquid chromatography , as described by Yamada and colleagues, using a Waters 510 HPLC pump operating at a flow of 1.8 ml/min, an Ultrasphere C18 column , and a Waters 996 Photodiodearray detector. Retinol and retinyl esters were monitored at 325 nm and quantified using standard curves relating the known mass to mass ratio of retinol or retinyl esters to that of internal standard retinyl acetate. This HPLC method allows for detection and quantitation of different retinyl esters, including retinyl palmitate, oleate, stearate, linoleate, and myristate.
Donât Miss: Hepatitis C Antibody With Reflex