Study Design And Population
This nationwide cohort study included patients infected with a nonepidemic HCV genotype treated with an interferon-free DAA regimen. Nonepidemic HCV genotypes were defined as genotypes and subtypes other than 1a/1b/2a/2b/3a/4a/4d. All laboratories performing HCV genotyping in the Netherlands were approached. All but 1 participated in the study: the Amsterdam University Medical Centers Sanquin Diagnostics, Amsterdam UMC Groningen, Groningen LUMC, Leiden Erasmus Medical Center, Rotterdam and Maastricht UMC, Maastricht.
Factors To Consider Prior To Choosing Retreatment Regimen
For retreatment of adults with HCV genotype 3 infection, several factors influence the regimen choice, including the prior regimen used when treatment failure occurred, the presence or absence of cirrhosis, and cost or insurance considerations. It is also worth noting that the clinical data for treatment-experienced individuals with HCV genotype 3 is more limited for the newest DAAs, such as glecaprevir-pibrentasvir, since these individuals have been encountered less frequently in recent years due to the efficacy of earlier DAA regimens. Therefore, the optimal duration of therapy for retreatment of persons with HCV genotype 3 with glecaprevir-pibrentasvir is not well established. The retreatment of individuals with HCV genotype 3 who have decompensated cirrhosis, renal impairment, acute HCV, or post-liver transplantation is not addressed in this lesson.
How Can You Identify What Genotype You Have
With the HCV infection, its important to know which genotype a person has. This will allow a healthcare provider to give the best care by creating a treatment plan specific to the type of HCV.
Overall, this is a relatively new component of HCV treatment. Before 2013, there wasnt a reliable way to distinguish between the different HCV genotypes that may be present in a person with the infection.
In 2013, the Food and Drug Administration approved the first genotyping test for people with HCV.
Various nucleic acid amplification tests can differentiate between the following genotypes:
- 1 and its subtypes
To do this, your doctor firsts obtain a sample of your blood plasma or serum. In the test, genetic material thats present inside the HCV virus is analyzed. During this time, several identical copies of complementary DNA material are produced. This testing can help identify the unique HCV genotype or genotypes present.
This test shouldnt be used as the first diagnostic tool for determining if a person has HCV infection.
However, anyone whos at risk for HCV should at least be tested for the disease with a screening test.
Don’t Miss: Dose Of Hepatitis B Vaccine Schedule
Generic Epclusa And Mavyret Arrive
Generic Epclusa
Then along came Epclusa, which is Sofosbuvir 400 mg + Velpatasvir 100 mg.Early trial results indicated that Epclusa was more effective against Hepatitis C G3 than Sofosbuvir + Daclatasvir or Sofosbuvir + Ribavirin but only very slightly, just 1% or 2%, which some statisticians would say was statistically insignificant.
The claim was that 12 weeks treatment of Epclusa was an effective treatment for Hep C genotype 3.
However I had heard this before and noticed that trials and real world treatments often give very different results.
In the real world, 12 weeks treatment with Epclusa gave about a 95% cure if the patient had not failed previous treatment.
But Epclusa was a great treatment option for G3 so when generic Epclusa became available I added it to the treatment options available for people with G3 and waited to hear the results.And now nearly 4 years after the release of generic Epclusa, the results are in Good news!
It would appear from a thorough study of all Genotype 3 treatment trial results that 12 weeks treatment with Epclusa will give about a 94% cure rate and 12 weeks with Sof + Daclatasvir will give about a 93% cure. Importantly longer treatment times will give significantly higher cure rates.
This graph shows that Epclusa is only 1% superior to Sofosbuvir + Daclatasvir except where a patient has failed previous treatment. When a patient with genotype 3 has failed previous treatment then Sof + Dac is superior.
Mavyret for Hep Genoptype 3
Assessments Of Efficacy And Safety
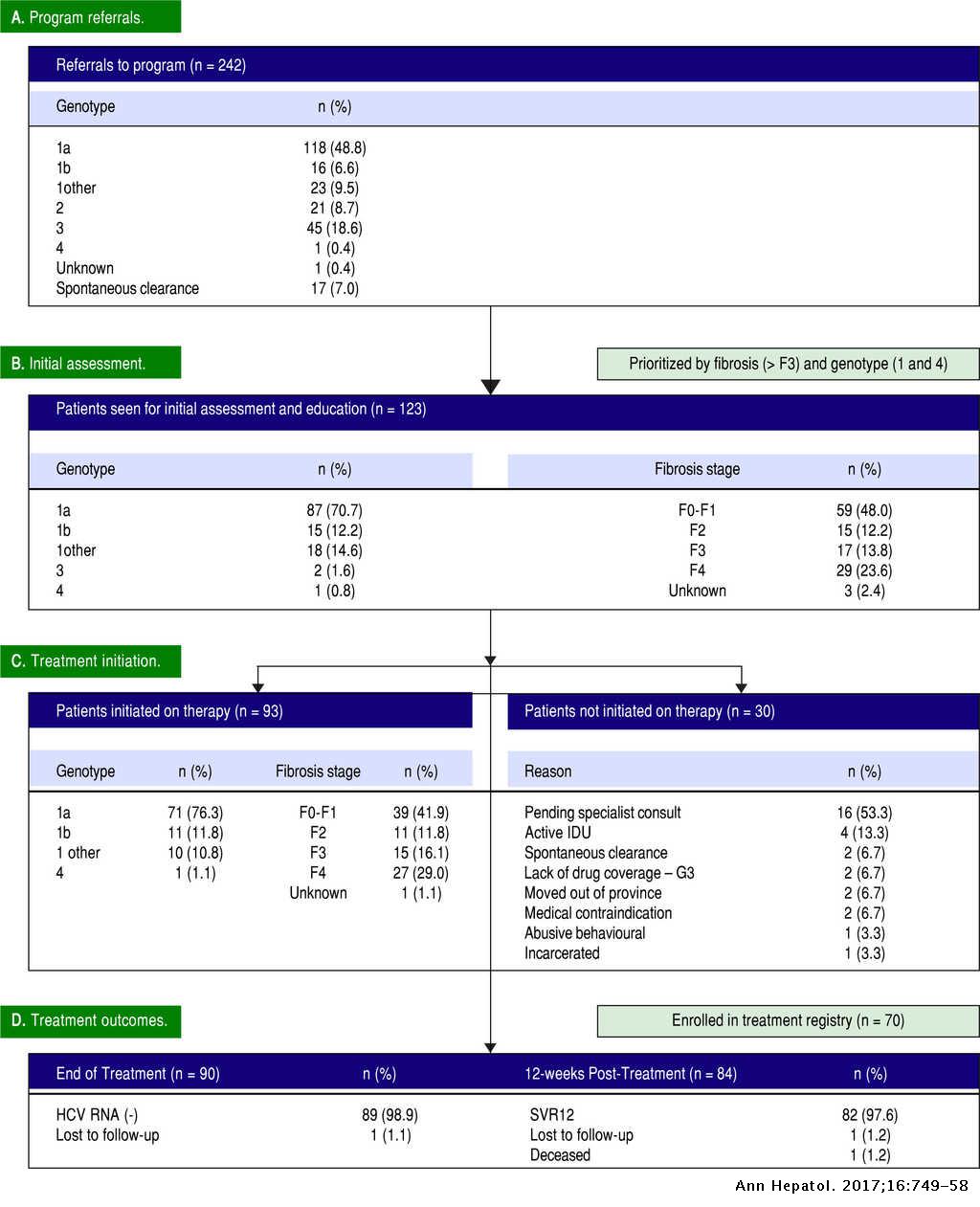
The patients were carefully monitored for clinical symptoms and adverse events including serious adverse events during the treatment and follow-up. Laboratory data including routine blood tests, biochemical liver and renal function and HCV RNA were regularly monitored at baseline, week 4 of therapy, the end of therapy , and at week 12 after the completion of therapy. Additional laboratory tests might be performed in some patients with the request of patient and the consultation of referring physician. Efficacy was measured by sustained virologic response at post-treatment week 12 . Serum HCV RNA was quantified by reverse-transcription polymerase chain reaction using the Cobas AmpliPrep/COBAS TaqMan HCV Test , which has a lower limit of 15IU/mL for HCV RNA quantification. HCV genotype was determined using RT-PCR with genotype-specific primers from the 5noncoding region of the virus. Routine blood tests and biochemical liver and renal functions were determined using standard procedures. The primary endpoint was SVR12, which was defined as serum HCV RNA undetectable at 12weeks after the end of therapy. The secondary endpoints were the treatment-related AEs and laboratory abnormalities.
Recommended Reading: How To Know If You Have Hepatitis B
Sofosbuvir Plus Pegylated Interferon/ribavirin
The combination of SOF plus PegIFN/RBV for 12 weeks is the only interferon based therapy recommended by the EASL and AASLD guidelines for the treatment of HCV genotype 3 infection .
In naive non-cirrhotic patients, SOF plus PegIFN/RBV for 12 weeks resulted in an overall SVR of 92-100% . However, efficacy data is scarce: few patients were included in clinical trials and only three studies evaluated the SVR rates in this population. The phase II study included 25 naive non-cirrhotic patients treated with SOF plus PegIFN/RBV for 12 weeks, reaching an overall SVR rate of 92%, but no SVR data according to specific genotype is available 70033-1.). Another phase II study included 17 patients treated with SOF plus PegIFN/RBV for either 12 or 8 weeks and the overall SVR rate was 100% in both arms . The Boson phase III study included 71 naive non-cirrhotic patients with HCV genotype 3 infection treated with SOF plus PegIFN/RBV for 12 weeks, achieving an overall SVR rate of 96% .
In non-cirrhotic patients, including naive and with previous failure to PegIFN/RBV, SOF plus PegIFN/RBV for 12 weeks resulted in high SVR rates . It must be noted that non-significant differences in SVR rates were observed among naive and treatment-experienced patients, but these data need to be cautiously analyzed, since only small cohorts were included in the studies.
Read Also: Is Hepatitis Ca Sexually Transmitted Disease
Hepatitis C Cure Rate Tops 90% In Hard
The sofosbuvir-based therapy was reviewed in a real-life Scandinavian study
Hepatitis C patients with hard-to-treat genotype 3 showed sustained virologic response of greater than 90% in a real-life study of a therapy based on the direct-acting antiviral drug sofosbuvir
HCV genotype 3 has been linked to increased risk of developing cirrhosis and hepatocellular carcinoma compared to other versions of the virus, previous studies have found.
Chronic hepatitis C virus genotype 3 infection with advanced liver disease has emerged as the most challenging to treat, researchers led by Olav Dalgard , MD, Ph.D. at Akershus University Hospital in Oslo, Norway noted in their study.
The research team from Norway, Denmark, Finland and Sweden chose patients from those four countries for the retrospective cohort study. About 100,00 people in Scandinavia are infected with HCV and half of them have genotype 3, according to the paper.
Genotype 3 is common all over Scandinavia and in the UK, Dalgard told MD Magazine in an email. We dont know why, but genotype 3 has its origin in South Asia and both the UK and Norway and Denmark have large immigrant populations from Pakistan.
Only a few studies have reported the effect of sofosbuvir-based treatment in genotype 3 patients with advanced liver disease in a real-life setting, the authors said.
Recommended Reading: Where Can I Get Hepatitis B Vaccine
You May Like: What’s Hepatitis B Vaccine
Interferons And Pegylated Interferons
The two most frequently used recombinant interferon preparations in clinical trials have been IFN alfa-2b and IFN alfa-2a , which differ from each other by only a single amino acid residue. IFN alfacon-1 , or consensus IFN, is a genetically engineered compound synthesized by combining the most common amino acid sequences from all 12 naturally occurring IFNs. Roferon-A was discontinued from the market in 2007 and Infergen was discontinued from the market in 2013.
The addition of propylene glycol molecules to IFN has led to the development of long-lasting IFNs that have better sustained absorption, a slower rate of clearance, and a longer half-life than unmodified IFN, which permits more convenient once-weekly dosing. The FDA has approved PEG-IFNs for the treatment of chronic hepatitis C.
Two PEG-IFN preparations are available for the treatment of chronic hepatitis C. PEG-IFN alfa-2b consists of IFN alfa-2b attached to a single 12-kd PEG chain it is excreted by the kidneys. PEG-IFN alfa-2a consists of IFN alfa-2a attached to a 40-kd branched PEG molecule it is metabolized predominantly by the liver.
Hepatitis C With Decompensating Cirrhosis
Until recently, doctors considered a liver transplant to be the only effective treatment for decompensating cirrhosis.
However, a recent small-scale study found that a course of direct-acting antiviral medication may improve some peoples liver function enough to take them off the waiting list for a liver transplant. People with liver disease that was less severe had a higher likelihood of removal from the list.
However, recent Canadian guidelines warn that certain antiviral drugs may potentially be dangerous for people with severe decompensating cirrhosis. This is because the liver is less able to filter out toxic waste, meaning that the antiviral drugs could accumulate to toxic levels. Doctors must weigh up the benefits against the risks.
When a person is waiting for a liver transplant, a doctor will assess whether or not to pause antiviral treatment.
Also Check: Can Hepatitis Turn Into Hiv
Epidemiology Of Hepatitis C Virus Genotypes And Genotypic Variations Around The World
HCV is divided into six distinct genotypes throughout the world with multiple subtypes in each genotype class. A genotype is a classification of HCV virus based on the genetic material in the RNA strands .
Figure 1.1.3. Genotypes of hepatitis C virus.
Genotypes 1, 2, and 3 have a worldwide distribution . Types HCV G1a and G1b predominate in Europe, North and South America, and Japan, accounting for about 60% of global infections. HCV G2 is common in Japan and China and is found in the United States and in northern, western, and southern Europe . HCV G3 is prevalent in Southeast Asia, India, and Australia and is variably distributed in Europe and the United States . HCV G4 is principally found in the Middle East, Egypt, and central Africa . HCV G5 is almost restricted to South Africa , and HCV G6 is found in specific regions in Asia .
Figure 1.1.4. Geographical distribution of hepatitis C virus genotypes.
Sofosbuvir Plus And Ribavirin
The combination SOF plus PegIFN/RBV for 12 weeks is recommended by EASL and AASLD for the treatment of naive or treatment-experienced patients with compensated cirrhosis and HCV genotype 3 infection . This recommendation is based in only one study, which observed an overall SVR rate of 8692% in compensated cirrhotic patients .
The Boson phase III study found SVR rates of 91% for the treatment of 23 naive patients with compensated cirrhosis with SOF plus PegIFN/RBV for 12 weeks . The treatment-experienced cirrhotic population was evaluated in two studies, which assessed the SVR rates with SOF plus PegIFN/RBV for 12 weeks . The Boson study included 35 patients and 30 achieved SVR . Lonestar, a phase II study, included 12 treatment-experienced cirrhotic patients and ten reached SVR .
Even though it was only evaluated in small cohorts, the regimen containing SOF plus PegIFN/RBV for 12 weeks presents an adequate option for patients with compensated cirrhosis however, interferon-based therapy may have some contraindications and is also associated with high rates of adverse events, especially in the cirrhotic population, justifying the ongoing search of safer and more effective therapies for these patients.
Read Also: What Are The Signs And Symptoms Of Hepatitis C
Treatment Of Hcv Genotype 3 Infection In Non
The combination of SOF plus RBV for 24 weeks was the first interferon-free therapy for patients with HCV genotype 3 infection approved by the FDA. International guidelines differ regarding the recommendations for this regimen. EASL guidelines do not recommend this therapeutic regimen for treatment-experienced cirrhotic patients. On the other hand, AASLD recommends SOF plus RBV as an alternative regimen for patients without cirrhosis with previous PegIFN/RBV failure or treatment-naive patients who are IFN-ineligible .
In naive non-cirrhotic patients with HCV genotype 3 infection, SOF plus RBV for 12 weeks resulted in an overall SVR of 61-68%. However, extending the treatment to 24 weeks led to an approximate 30% increase in SVR rates, ranging from 90 to 96% .
Two large clinical trials evaluated the efficacy of SOF plus RBV for 12 weeks in naive, non-cirrhotic patients infected with HCV genotype 3. The Fission trial included 145 patients, but only 89 achieved SVR . The Positron trial included 84 naive patients who were interferon-ineligible or intolerant, of which 57 reached SVR . The Boson clinical trial found higher SVR among naive and non-cirrhotic patients treated with SOF plus RBV for 16 weeks. Among 70 patients treated, 58 achieved SVR . Another arm of this study evaluated 72 naive non-cirrhotic patients treated for 24 weeks, with an overall SVR of 90% . SOF plus RBV for 24 weeks was also used in the Valence trial, which included 92 patients and 87 achieved SVR .
Treatment With Sof + Dcv
SOF + DCV had been administered to 193/316 patients among whom 123 also received RBV. Treatment duration was short in 119/193 and long in 74/193 patients .
In the mITT group, SVR12 was obtained in 177/193 of those treated with SOF + DCV +/-RBV. SVR12 rates did not differ between those who received shorter versus longer treatment duration and 66/74 respectively. Nor did it differ between those who did and did not receive RBV versus 62/70 , respectively). However, among those who received short treatment there was a trend towards higher SVR12 rates in patients who received RBV compared with those who did not and 33/38 respectively p = 0.055). In further bivariate analyses, SVR12 was independent of age, gender, treatment experience, the presence of cirrhosis, the presence of decompensated liver disease and liver elasticity.
SOF+DCV was administered to 20 patients with Child Pugh B or C and SVR was achieved in 17 of these. Treatment duration was 24 weeks in 15 and 1216 weeks in 5 patients. RBV was administered to 12 patients.
Don’t Miss: Hepatitis C Causes Liver Cancer
Significance Of The Study
What is already known about this subject?
-
Hepatitis C virus genotype-3a is now the most prevalent HCV subtype in the UK and South Asia.
-
Prophylactic and therapeutic vaccines are currently in development against genotype-1 infection. However, the T cell targets in genotype-3 infection are currently unknown.
-
Interferon therapy has immunomodulatory properties HCV genotype-3a is more responsive to interferon based therapies than HCV genotype-1. The effects of interferon on genotype-3a T cell immunity are not known.
-
IL28B linked polymorphisms are associated with sustained virological response rates in HCV genotype-1 but not genotype-3a infections therefore, alternative mechanisms that explain this observation should be explored.
What are the new findings?
-
In contrast to genotype-1 infection, genotype-3a-specific CD8 T cell responses commonly target the non-structural hepatitis C virus proteins in chronic disease.
-
In chronic genotype-3a infection, T helper responses target a dominant HCV core protein.
-
T cell targets identified during chronic infection may have a limited role in protective immunity since these differ from those found in spontaneously resolved infection where CD4 T cells targeting non-structural proteins dominate.
-
Paradoxically, genotype-3a-specific T cell responses and total lymphocyte counts decline during interferon treatment in association with a sustained virological response.
How might it impact on clinical practice in the foreseeable future?
Hyperlipidemia Diabetes Mellitus Or Ir
Lipid metabolism is intimately involved in the molecular mechanisms of the HCV infectious cycle. HCV replication influences and depends upon cholesterol uptake and efflux through different lipoprotein receptors during its entry into the hosts cells . Very low-density lipoprotein-associated proteins, including apolipoprotein B, apoE and microsomal triglyceride transfer proteins, have been shown to play a crucial role in the formation of infectious HCV particles, especially pertaining to genotype 3 . HCV can bind low-density lipoprotein receptors and lead to intracellular lipid deposition . Patients infected with genotype 3 tend to have hypocholesterolemia and hypobetalipoproteinemia, which may account for the direct effect of the virus on lipid metabolism . It appears that HCV-3 also selectively interferes with the late cholesterol pathway, a phenomenon that appears to disappear with SVR . Interestingly, high LDL levels tends to predict SVR in patients treated with interferon and ribavirin . Since LDL receptors are involved in HCV entry into hepatocytes, higher LDL levels may decrease the number of LDL receptors on the cell membrane, thus decreasing cellular infectivity .
Also Check: How To Cure Hepatitis B
Why Do Genotypes Matter For Treatment
Knowing your HCV genotype is important information that can help patients and doctors find the most effective treatment.
All HCV genotypes cause the same amount of liver damage. However, people infected with genotype 1, particularly subtype 1b, may have a greater chance of developing cirrhosis, or severe liver scarring, than other genotypes. Genotypes 1b and 3 may increase the risk of liver cancer.
HCV can now be cured by all oral, direct-acting antivirals , medications that prevent the hepatitis C virus from make copies of itself. DAAs do this by sticking to proteins in the virus and blocking steps in the virus life cycle. This allows your immune system to clear the virus out of your body. How well a DAA works depends on where it sticks to the target proteins in the virus.
Some of the latest DAA treatments are pangenotypic, which means they can cure all genotypes at nearly the same rates.