Search For Signs Of Hepadnaviral Coinfection
Because human HDV transmission requires an envelope protein provided by an active coinfection with HBV, we subjected all 763 P. semispinosus blood samples to PCR screening for hepadnaviruses. The assay was designed to detect all mammalian orthohepadnaviruses, including those from rodents, bats, and primates . None of the samples tested positive. Specific reanalysis of all available transcriptome datasets from the present study did not yield any evidence for orthohepadnaviral genomes in a basic local alignment search tool analysis. All blood samples with sufficient volume, whether RNA-positive or -negative for RDeV , were tested for antibodies against woodchuck HBV core antigen . This antigen was chosen because the woodchuck is most closely related to Proechimys among known HBV hosts. In previous studies, we demonstrated that anti-HBc antibodies broadly cross-react among HBVs from different hosts, even across mammalian orders , so it is likely that WHcAg would be bound by antibodies against a possible HBV in Proechimys. However, no such antibodies were found in any of the tested samples .
Certain Infectious And Parasitic Diseasesincludes
- certain localized infections – see body system-related chapters
- carrier or suspected carrier of infectious disease
- infectious and parasitic diseases complicating pregnancy, childbirth and the puerperium
- infectious and parasitic diseases specific to the perinatal period
- influenza and other acute respiratory infections
- code to identify resistance to antimicrobial drugs
- herpesviral hepatitis
- 2016201720182019202020212022Non-Billable/Non-Specific Code
Includes
- Chronic hepatitis b with hepatic coma
- Chronic type b viral hepatitis
- Hepatitis b, chronic
- Hepatitis b, chronic, with hepatic coma
- Inflammation of the liver in humans caused by hepatitis b virus lasting six months or more. It is primarily transmitted by parenteral exposure, such as transfusion of contaminated blood or blood products, but can also be transmitted via sexual or intimate personal contact.
- 441 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis with mcc
- 442 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis with cc
- 443 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis without cc/mcc
- 791 Prematurity with major problems
- 793 Full term neonate with major problems
- : New code
- 2017
Listing Of Usual Initial Therapeutic Options Including Guidelines For Use Along With Expected Result Of Therapy
Goals of antiviral therapy are HBeAg seroconversion , HBsAg loss, and suppression of HBV DNA. All patients with cirrhosis should continue treatment indefinitely. Patients without cirrhosis can consider discontinuation after 12 months of achieving above mentioned goal. However, the risk for seroconversion or recurrent viremia persists and patients need to be monitored every 3 months for at least one year after discontinuation of antiviral therapy.
Also Check: How Hepatitis C Virus Spread
Investigation Of Hepacivirus As Potential Cofactor
Experimental evidence shows that apart from HBV, other viruses, including hepatitis C virus , can provide envelope proteins for human HDV . As we previously detected a high prevalence of hepacivirus in the rodents studied , this possibility was investigated. Overall, we found 4 out of 30 RDeV RNA-positive rodents in which hepacivirus was not detected by the tests initially applied . To exclude having missed hepacivirus detection, we applied RT-PCR assays specifically designed for the E1, NS3, and NS5B genes of P. semispinosus hepacivirus to those samples. All four animals were confirmed to be hepacivirus RNA-negative by these assays as well as by RNA-seq read mapping against P. semispinosus hepacivirus. Statistical analysis of the degree of dependency between deltavirus and hepacivirus infection in individuals was conducted, providing no support for a dependency between the two infections . Also, analyses of sampling site-specific deltavirus and hepacivirus detection rates did not reveal any correlation between deltavirus and hepacivirus , arguing against a linear correlation. Logarithmic and linear regression fits did not yield significant associations either, suggesting that the two viruses follow different patterns of distribution. We conclude that RDeV infection in P. semispinosus does not depend on active hepacivirus infection.
How Can I Be Sure That The Patient Has Hepatitis B Virus Infection
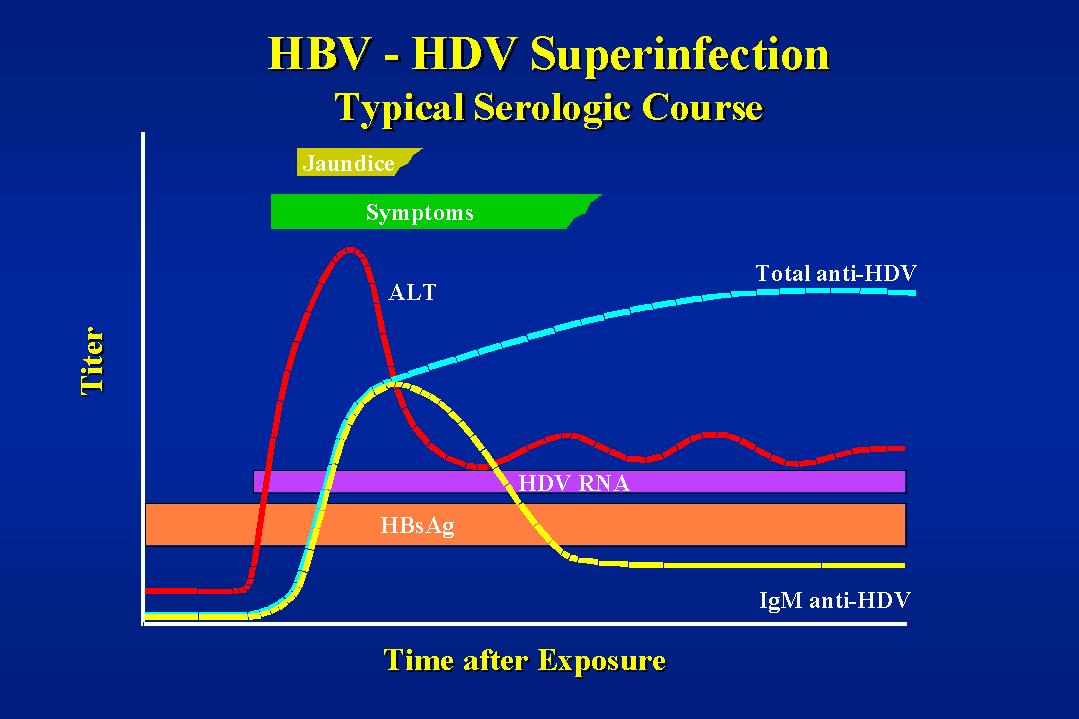
HBV infection presents with nonspecific features. Infection with HBV has a wide spectrum of manifestations, including subclinical hepatitis, anicteric hepatitis, icteric hepatitis, and fulminant hepatitis during the acute phase and the asymptomatic carrier state. HBV infection includes chronic hepatitis, cirrhosis, and hepatocellular carcinoma during the chronic phase.
Approximately 70% of patients with acute HBV infection have subclinical hepatitis or anicteric hepatitis, whereas 30% become icteric. Acute liver failure develops in approximately 0.1% to 0.5% of patients. The incubation period lasts 1 to 4 months. A serum sickness-like syndrome may develop during the prodromal period. This is followed by constitutional symptoms such as low-grade fever, malaise, anorexia, nausea and vomiting, and right upper quadrant or midepigastric pain. Jaundice usually appears as the constitutional symptoms begin to subside. Clinical symptoms and jaundice generally disappear after 1 to 3 months, but some patients may have prolonged fatigue, even after normalization of aminotransferase levels.
Read Also: How Would You Know If You Had Hepatitis C
A Tabular Or Chart Listing Of Features And Signs And Symptoms
HBV or HDV infection does not have any pathognomonic or characteristic features.
Some less common clinical presentations
Extrahepatic manifestations occur in approximately 10% of patients with chronic HBV infection. Mediated by circulating immune complexes, acute hepatitis may be heralded by a serum sickness-like syndrome manifested as fever, skin rashes, arthralgia, and arthritis, which usually subside with the onset of jaundice.
Hepatitis B virus-related glomerulonephritis, most commonly membranous glomerulonephritis, is another manifestation.
Other extrahepatic manifestations include rarely essential mixed cryoglobulinemia and aplastic anemia.
Other diseases and conditions that might mimic the signs, symptoms, or clinical features of HBV infection
Other viral hepatitis , perform HCV antibodies, and HCV RNA.
What Other Diseases Conditions Or Complications Should I Look For In Patients With Hepatitis B Virus Infection
Acute HBV infection can lead to fulminant hepatitis. Acute liver failure develops in approximately 0.1% to 0.5% of patients.
Prothrombin time, which reflects hepatic synthetic function, is the best indicator of prognosis.
Progression to cirrhosis occurs in up to 20% of patients with untreated chronic hepatitis B. Annual incidence of hepatic decompensation is 5-8% and hepatocellular carcinoma is 2-4% in patients with cirrhosis. Rates of cirrhosis and HCC increase substantially with HBV DNA level, alt levels, HBeAg positivity, and genotype C. Additional risk factors include advanced age, male gender, immunocompromised state, concomitant viral infection , alcohol use, and metabolic syndrome. Furthermore, risk of HCC is higher in patients from Sub-Saharan Africa, positive family history, and smoking.
Read Also: Can You Donate Plasma If You Had Hepatitis B
Age And Sex Distribution
Historically, notification rates have consistently been highest in young adults aged 1519 years, 2024 years and 2529 years . Since 2001, notification rates in these three age groups have declined, particularly in 1519 year olds, where for the first time in 2005 rates fell below 1 per 100,000 to 0.8 per 100,000. Rates have remained fairly stable in the other age groups from 1993 to 2005. As in previous years, there were more male than female notifications in almost all age groups in 2003, 2004 and 2005, with an overall male:female ratio of 1.7:1.
During the period 2002/2003 to 2004/2005, rates for hospitalisations with a principal diagnosis of acute hepatitis B were highest in adults aged 2529 years and 2024 years . Like notifications, and as in previous years, hospitalisations occurred predominantly in males with an overall male:female ratio of 1.8:1.
Figure 8. Acute hepatitis B notification rates, Australia, 1993 to 2005,* by age group
* Notifications where the month of diagnosis was between January 1993 and December 2005.
Figure 9. Acute hepatitis B hospitalisation rates, Australia, 2002/2003 to 2004/2005,* by age group and sex
* Hospitalisations where the principal diagnosis was acute hepatitis B and the month of separation was between 1 July 2002 and 30 June 2005.
Organ Distribution Of Rdev Rna And Protein Detection
To obtain insight into the infection pattern caused by the rodent deltavirus, we tested for viral RNA in organs of 18 animals that were found dead during sampling . One of these animals tested RDeV RNA-positive in all available organs , but its organ-specific RNA concentrations did not suggest specific virus replication in the liver . Also, a commercial HDV antigen enzyme-linked immunosorbent assay that we found to cross-detect with RDeV did not identify organ-specific protein expression in the liver, lung, small intestine, heart, or kidney of this animal . To further understand potential virus excretion, fecal samples from 822 individuals were tested by RT-PCR, 10 of which were positive for RDeV RNA . This 1.2% detection rate was significantly lower than that in blood samples . The average virus concentration in fecal samples was 2.9 × 109 RNA copies per gram, with a maximum concentration of 2.2 × 1010 copies per gram. Average and maximum concentrations in blood were 1.8 × 108 and 2.1 × 109 copies per milliliter.
You May Like: How Many Hepatitis Are There
How Can I Confirm The Diagnosis
What tests should be ordered first?
BsAg is the hallmark of HBV infection. It is usually detectable 1 to 10 weeks after an acute exposure to HBV, and approximately 2 to 6 weeks before the onset of clinical symptoms. Most patients who recover from acute hepatitis B clear HBsAg in 4 to 6 months. Persistence of HBsAg in serum for more than 6 months implies chronic infection. Anti-HBs can be detected in individuals who recovered from HBV infection and in those who successfully responded to hepatitis B vaccination.
The antigen HBeAg is a marker of HBV replication and infectivity. Its presence is usually associated with the detection of HBV DNA in serum and a high risk of transmission of infection. However, HBeAg-negative and anti-HBe-positive patients may continue to have active liver disease and moderate levels of HBV DNA in serum. These patients usually have HBV variants with diminished or abolished production of HBeAg.
In patients with acute hepatitis B, serum HBV DNA appears early and may precede the detection of HBsAg.
Chronic hepatitis B is categorized into four phases based on the virus activity and the patientâs immune response to the virus activity . Patients may evolve through some or all phases with variable duration.
-
Inactive carrier: HBeAg , HBV DNA low, ALT normal
-
Immune tolerant: HBeAg , HBV DNA elevated, ALT normal
-
Immune active: HBeAg , HBV DNA elevated, ALT elevated
-
Immune reactivation: HBeAg , HBV DNA elevated, ALT elevated
A Listing Of A Subset Of Second
There are currently six medications approved for use in the United States. Choice of antiviral agent is driven by side effect profile, co-morbidities, prior therapy exposure, HBV genotype, costs, and pregnancy state.
Peg-IFN-2a: dose of 180 mcg weekly. Side effects include flu-like symptoms, mood disturbances, cytopenias, autoimmune disorders. Monitoring should include a CBC and TSH every 3 months, monitoring for infection, autoimmune disorders, neuropsychiatric complications, and infections. Pregnancy category C.
Entecavir: dose of 0.5 or 1.0 mg daily. Side effects include lactic acidosis. Pregnancy category C.
Tenofovir: dose of 300mg daily. Side effects include nephropathy, Fanconi-like syndrome, osteomalacia, lactic acidosis. Monitoring should include yearly creatinine clearance, serum phosphate, urine glucose and protein annually, as well as bone density scan if at risk. Pregnancy category B.
Lamivudine: dose of 100mg daily. Side effects include pancreatitis, lactic acidosis. Monitoring should include creatinine kinase and lactic acid if clinical concerns. Pregnancy category C.
Telbivudine: dose of 600mg daily. Side effects include creatine kinase elevations, myopathy, peripheral neuropathy, and lactic acidosis. Monitor creatinine kinase if symptoms. Pregnancy category B.
Recommended Reading: Pictures Of Hepatitis Skin Rashes
Infectious Diseasechronic Disease Connections
For centuries, physicians and scientists hypothesized that infection might explain some chronic syndromes. Proof, however, lagged behind speculation. A paucity of tools to detect many agents and the challenges of linking past infectionsometimes decades in the pastwith present chronic illness perpetuated the idea that most infectious diseases are acute illnesses, and that chronic diseases have noninfectious causes. By the latter third of the 20th century, however, exceptions to this dogma began to emerge. For example, hepatitis B virus infection came to explain a large proportion of chronic liver disease and hepatocellular carcinoma in areas of endemic infection . However, it was the discovery that Helicobacter pylori can induce gastric inflammation that truly transformed conventional thinking about the noncommunicable nature of many chronic conditions in recognition of this groundbreaking achievement, Marshall and Warren were awarded the Nobel Prize in Physiology or Medicine 2005. Researchers have subsequently demonstrated that eradication of H. pylori can cure most cases of peptic ulcer disease, a chronic condition long attributed to noninfectious factors such as stress, diet, smoking, and family history . Today, scientists and physicians widely recognize the plausibility of infectious agent origins for chronic diseases.
Figure 1
Figure 1. Emergence timeline for infectious determinants of chronic diseases.
Severe Morbidity And Mortality
For patients with a principal diagnosis of acute hepatitis B, 2,408 hospital bed days were recorded. The median length of stay was three days, with longer stays for adults aged 60 years and over . There were 22 deaths from acute hepatitis B recorded in the two years 2003 to 2004, 17 in males and five in females. All of the deaths occurred in those aged 25 years and over, and nearly two thirds were aged over 60 years, in whom nine of the fourteen were males. In 2003, there were twice as many deaths as in 2004 . There were four cases of hepatic coma recorded among hospitalisations with a principal diagnosis of acute hepatitis B, with none of these cases recorded as having delta co-infection . There were three deaths reported to NNDSS in notified cases between 2003 and 2005.
Table 5. Acute hepatitis B notifications, hospitalisations and deaths, Australia, 2002 to 2005,* by age group
| Age group |
|---|
| 4 | 0.8 |
* Measured using National Hospital Morbidity data where the month of hospital separation was between 1 July 2002 and 30 June 2005.
ICD-10-AM codes B16.0 and B16.2.
Recommended Reading: How Do You Say Hepatitis C In Spanish
What Is The Right Therapy For The Patient With Hepatitis B Virus Infection
Treatment with antiviral therapy is recommended in patients with chronic hepatitis B in the immune active phase with elevated HBV DNA levels and ALT greater than 2x the ULN. Patients with cirrhosis and HBV DNA > 2,000 IU/mL should be treated regardless of ALT levels. If patients do not meet these cutoff criteria, antiviral therapy should still be considered if older age, positive family history, presence of extrahepatic manifestations, or prior treatment).
Treatment with antiviral therapy is generally not recommended for patients with immune tolerant chronic hepatitis B. However, patients should have labs checked every 6 months to look for evidence of activation. Additionally, in spite of normal ALT levels, patients should be treated with antiviral therapy if there is evidence of necroinflammation or fibrosis.
What treatment options are effective?
Several agents are currently approved for the treatment of chronic hepatitis B: interferon alpha , pegylated interferon alfa 2a, lamivudine, adefovir, entecavir, telbivudine, and tenofovir. Each agent has inherent limitations.
With the drugs currently available, the physician may consider two different concepts for the treatment of chronic hepatitis B: the first concept is that of sustained response obtained after a limited duration of therapy with pegylated interferon the second concept is that of maintained response obtained during prolonged administration of therapy with analogues.
How Should I Monitor The Patient With Hepatitis B Virus Infection
Prevention through universal vaccination has effectively decreased the incidence of liver cancer, and new therapeutic agents may delay or avoid the establishment of cirrhosis. All patients with cirrhosis should be screened for HCC with ultrasound every 6-12 months. Additionally, some high risk populations with chronic hepatitis B should be screened even in the absence of cirrhosis. These populations include Asian men over 40 years old, Asian women over 50 years old, African men and women over 20 years old, persistent ALT elevation, HBV DNA level > 2,000 IU/mL, and patients with a family history of HCC.
The only chance for long-term survival after HCC diagnosis is to achieve early detection through regular surveillance by ultrasound and AFP determination. This will allow the indication of effective therapy such as surgical resection, liver transplantation or percutaneous ablation.
Don’t Miss: Can You Catch Hepatitis C Through Sex
Chronic Viral Hepatitis B Without Delta
- 2016201720182019202020212022Billable/Specific Code
- B18.1 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes.
- The 2022 edition of ICD-10-CM B18.1 became effective on October 1, 2021.
- This is the American ICD-10-CM version of B18.1 – other international versions of ICD-10 B18.1 may differ.
- Carrier of viral hepatitis B
- Chronic hepatitis B
- Applicable To annotations, or
Deltavirus Antigens And Editing Of The Amber Codon
One key feature of HDV is the expression of two viral proteins from a single ORF. A 19-amino acid carboxyl-terminal extension differentiates the S-HDAg from the L-HDAg in human HDV. This extension is the result of antigenomic editing by cellular adenosine deaminase acting on RNA 1 at a site corresponding to the amber stop codon in the S-HDAg ORF of the circular antigenomic HDV RNA, edited from a UAG to a UIG codon. Following HDV RNA replication, the UIG codon is converted into a tryptophan codon during subsequent HDV messenger RNA synthesis, allowing expression of the L-HDAg . Interestingly, a 19-amino acid carboxyl-terminal extension is present in RDeV, though different in 15 of the 19 amino acids. To investigate potential RDeV large antigen expression, the full RDeV genome was cloned as a tandem head-to-tail fusion construct in genomic orientation downstream of a CMV promoter and transfected into HuH7 cells. As positive controls, S- or L-RDeAg was expressed under the control of a CMV promoter. Specific S- or L-RDeAg expression was examined with antibodies from rabbits immunized with synthetic peptides against S-RDeAg or the putative 19-amino acid extension of L-RDeAg . The tandem genome construct expressed S-RDeAg but not L-RDeAg .
Read Also: Is Hiv The Cause Of Hepatitis B