Approach To Choosing Hcv Genotype 1 Treatment Regimen
For individuals with chronic HCV genotype 1 infection, the main factors that influence the choice and duration of therapy are cirrhosis status and prior treatment experience. With the use of certain regimens for persons with HCV genotype 1a, namely elbasvir-grazoprevir, the genotype 1 subtype also impacts the choice of therapy, as elbasvir-grazoprevir is only recommended for persons with HCV genotype 1a who do not have baseline NS5A resistance-associated substitutions . In addition, the HCV RNA level and the patients HIV status can impact the duration of ledipasvir-sofosbuvir, but does not affect the duration of other regimens. Finally, the cost of the regimen, insurance coverage, and provider preference can play a major role in the regimen choice. The following treatment recommendations are based on the AASLD-IDSA HCV Guidance for initial treatment of adults with HCV genotype 1 and for retreatment of adults in whom prior therapy failed, including those with HCV genotype 1.
Genotype Table: Recommended Treatments For Hepatitis C Genotypes 1 2 3456
| Generic Brand Name | |||||
|---|---|---|---|---|---|
| Hepatitis c Genotype 1, 4, 5, 6 treatment. It is mainly used to treat Hep C | A combination pill taken once a day. Its usually taken for 12-24 weeks, and sometimes only 8 weeks. | Major side effects are rare. | Natco, Hetero, Mylan, Zydus Cadila, Dr. Reddy | ||
| Hepcinat, Sofovir, Myhep, Sovihep, Resof | 400 mg Sofosbuvir | 2, 3, 4, 5, 6 | 1 Sofosbuvir pill and 1 Daclatasvir pill once a day for 12 weeks. Sometimes used with Ribavirin. | Major side effects are rare. | Natco, Hetero, Mylan, Zydus Cadila, Dr. Reddy |
How Genotypes Affect Treatment
Medications known as direct acting antivirals, or DAAs, stop the hep C virus from making copies of itself. Some DAAs appear to work well on all hepatitis C genotypes. Others work on only one or some.
Your doctor will probably prescribe some combination of these medications:
- Velpatasvir
Some pills combine two drugs into one pill.
You’ll probably take these meds for anywhere from 8 to 12 weeks. But they may not be right for everyone because of things like cost or other illnesses.
Your specific genotype can tell your doctor important things about how to use those medications, what to watch for, and other drugs you might need.
For example, you may have a higher chance for cirrhosis if you have genotype 1.
Genotype 3, the second most common subtype worldwide, may not respond as well to DAAs alone. In addition, this type might suggest that:
- Liver cancer is more likely.
- Insulin resistance might happen. When your body resists or doesn’t respond to insulin as well as normal, you have a higher chance of heart disease and diabetes.
- You might need longer, more challenging treatment
Your doctor might adjust or change your DAA treatment if you have:
Show Sources
American Association for the Study of Liver Diseases: âInitial Treatment of Adults with HCV Infection,â âHCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C.â
CDC: âHepatitis C Questions and Answers for the Public.â
Infohep.org: âHepatitis C treatment factsheet: Harvoni .â
Recommended Reading: How Is Hepatitis C Contracted
Genotypes And Hcv Genetic Heterogeneity As Epidemiologic Markers
Because of geographic clustering of distinct HCV genotypes, genotyping may be a useful tool for tracing the source of an HCV outbreak in a given population. Examples include tracing the source of HCV infection in a group of Irish women to contaminated anti-D immunoglobulins . All of these women were infected with HCV genotype 1b, a genotype identical to the isolate obtained from the implicated batch of anti-D immunoglobulin. Hohne et al. used genotyping to trace the sources of outbreaks in Germany . More recently, genotyping and molecular characterization of HCV isolates provided evidence for a patient-to-patient transmission of HCV during colonoscopy . The index case as well as the two other infected patients had HCV genotype 1b. Nucleotide sequencing of the NS3 region showed that the three patients had the same isolate , strongly suggesting a common source of infection.
Although Zein et al. found no association between HCV genotypes and the mode of HCV acquisition in their population, others have provided evidence for such an association . It has been suggested that genotypes 3a and 1a are closely associated with intravenous drug use and that genotype 1b is seen more often in patients who acquired HCV through blood transfusion. This information may be useful in tracing sources of HCV epidemics.
Learning Objective Performance Indicators
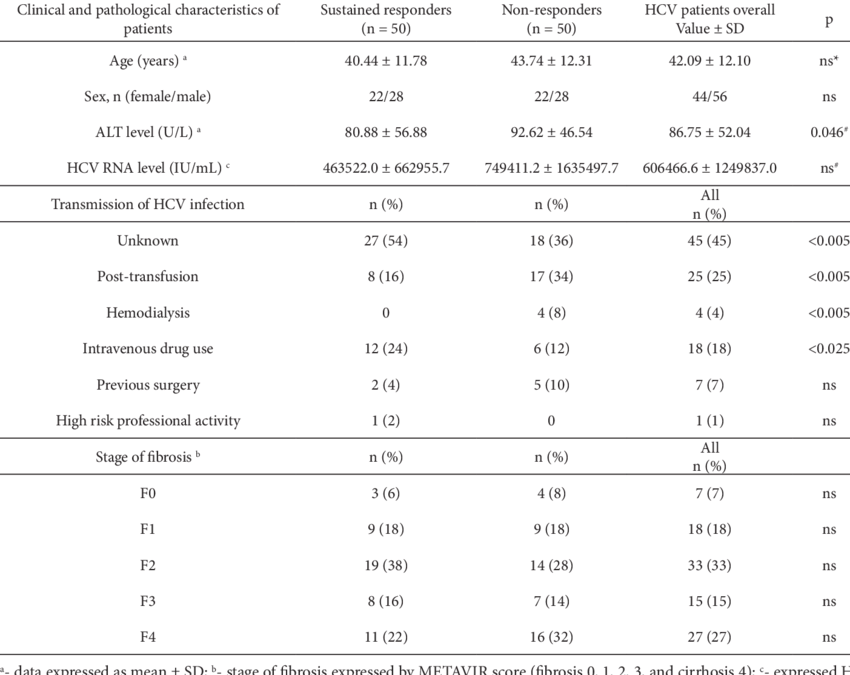
- List key factors that influence the choice and duration of therapy for treating HCV genotype 1 infection
- Describe the circumstances for NS5A genotypic drug resistance testing
- Discuss preferred therapies for initial treatment of treatment-naïve adults with HCV genotype 1 infection
- List the preferred therapies for retreatment of adults with chronic HCV genotype 1 infection who have previously failed therapy
Also Check: What Is Hepatitis B Vaccine For
Geographic Distribution Of Hcv Genotypes
At least six major genotypes of HCV, each comprising multiple subtypes, have been identified worldwide . Substantial regional differences appear to exist in the distribution of HCV genotypes .2). Although HCV genotypes 1, 2, and 3 appear to have a worldwide distribution, their relative prevalence varies from one geographic area to another .2).
Worldwide geographic distribution of HCV genotypes and subtypes. Others indicate unclassified sequences.
HCV subtypes 1a and 1b are the most common genotypes in the United States 3) . These subtypes also are predominant in Europe . In Japan, subtype 1b is responsible for up to 73% of cases of HCV infection . Although HCV subtypes 2a and 2b are relatively common in North America, Europe, and Japan, subtype 2c is found commonly in northern Italy. HCV genotype 3a is particularly prevalent in intravenous drug abusers in Europe and the United States . HCV genotype 4 appears to be prevalent in North Africa and the Middle East , and genotypes 5 and 6 seem to be confined to South Africa and Hong Kong, respectively . HCV genotypes 7, 8, and 9 have been identified only in Vietnamese patients , and genotypes 10 and 11 were identified in patients from Indonesia . There has been disagreement about the number of genotypes into which HCV isolates should be classified. Investigators have proposed that genotypes 7 through 11 should be regarded as variants of the same group and classified as a single genotype, type 6 .
Antiviral Treatment In Pregnancy
The risk of vertical transmission in mothers with an HCV monoinfection is 5% and is not diminished by cesarean section . Mothers are not advised against breastfeeding either. Vertically acquired hepatitis C takes a mild course in childhood, with very slow progression of hepatic fibrosis . Antiviral treatment during pregnancy cannot be recommended, as there are insufficient data on the potential teratogenicity of DAAs.
Recommended Reading: How Long Does Hepatitis C Live Outside The Body
Model Structure And Assumptions
In this study, an established Markov model , developed in Microsoft Excel 2013, was used from Chinese payer perspective to estimate the lifetime outcomes of treating a cohort of HCV genotype 1b treatment-naïve patients with two hypothetical regimens, DCV+ASV and PR . The model runs in annual cycles over a lifetime horizon and an annual discount rate of 5% was applied.
Genomic Organization Of Hcv
The original isolate was a positive-sense RNA virus with approximately 9,400 ribonucleotides, containing a poly tail at the 3 end .1). The sequence contained a 5 untranslated region of 341 bases, a long open reading frame coding for a polyprotein of 3,011 amino acids, and a 3 untranslated region of about 27 bases. This RNA structure is most similar to that of the family Flaviviridae, which encompasses numerous arthropod-borne viruses. Consistent with the known functions of most flavivirus proteins, the three N-terminal HCV proteins are probably structural and the four C-terminal proteins are believed to function in viral replication.
Genomic organization of HCV. First generation, second generation, and third generation refer to serologic assays for detection of HCV antibodies.
The open reading frame length of each genotype is characteristically different. Whereas the open reading frame in type 1 isolates is approximately 9,400 ribonucleotides, that of type 2 isolates is typically 9,099 nucleotides and that of type 3 isolates is typically 9,063 nucleotides . These differences may potentially account for some of the phenotypic differences among genotypes discussed below.
You May Like: Can You Get Hepatitis C From Drinking After Someone
Different Types Of Hcv Genotypes:
There are generally six main kinds of genotypes of hepatitis C virus genotype 1, 2,3,4,5 and 6. These include many subtypes. Underdeveloped countries are mostly affected by Genotype 4 and 5. Genotype 4 is most commonly found in the Middle East and Africa. United States is mostly affected by Hepatitis C genotype 1. Hepatitis C Genotypes 1,2 and 3 are found throughout the world. Genotype 5 is common in South Africa. Asians are mostly affected by Genotype 6.
Chemistry Mechanism Of Action Spectrum And Resistance
Simeprevir is an inhibitor of the NS3/4A serine protease of HCV, which is necessary for the proteolytic cleavage of the HCV-encoded polyprotein and is essential for viral replication. Simeprevir has demonstrable in vitro activity against HCV genotypes 1a, 1b, and 4 using different cell-based replicon assays.
Several amino acid substitutions at NS3 protease positions confer reducted activity, including F43, Q80, S122, R155, A156, and D168. The baseline prevalence of Q80K is 30% in genotype 1ainfected patients, but is less than 1% in genotype 1binfected patients. The Q80K polymorphism is associated with a lower sustained virologic response in clinical trials. Baseline determination of the Q80K polymorphism should be obtained before initiation of treatment with simeprevir.
ELIZABETH I. HARRIS, … LAURA W. LAMPS, in, 2009
You May Like: Can You Donate Blood If You Had Hepatitis A
What Is The Current Research Into Genotypes And Treatments For Each Type
The most widely used anti-HCV therapy, PEG/ribavirin, doesnt target the virus itself. This treatment regimen primarily affects the persons immune system. Its goal is to rally the immune system to recognize and eliminate cells infected with HCV.
However, variations of HCV in a single person wont necessarily look the same to the immune system. This is one of the reasons that HCV infections persist and become chronic infections.
Even with this genetic diversity, researchers have identified proteins that are required for the reproduction of HCV in the body. These proteins are present in essentially all of the many HCV variants.
The new treatments for HCV target these proteins. That means they target the virus. Direct-acting antiviral therapy uses small molecules designed to specifically inhibit these viral proteins.
Many DAA drugs have been under development during the past decade. Each drug targets one of the handful of essential HCV proteins.
The first two DAA drugs, boceprevir and telaprevir, got approval for use in the United States in 2011. Both target a particular type of HCV enzyme known as protease. These drugs are used in combination with PEG/ribavirin.
Both of these new medications are most effective for HCV genotype 1. Theyre moderately effective for genotype 2, and not effective for genotype 3.
Initially, they were only approved for use in people with genotype 1 HCV in combination with PEG/ribavirin.
Why Do People Have Different Genotypes
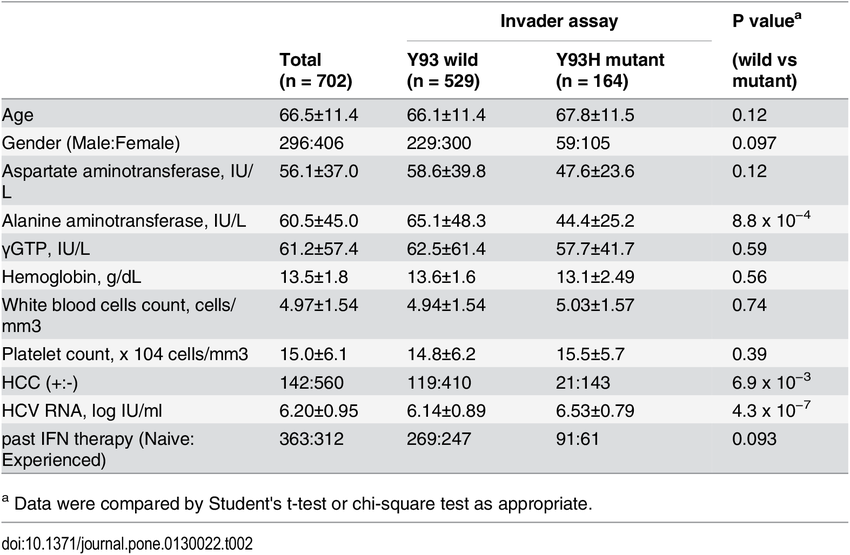
A person of any racial or ethnic group can carry any genotype or subtype. However, some may be more prevalent in some racial or ethnic groups than others. In the United States, over 90% of African Americans, compared to 67% of Caucasians, carry genotype 1.
People who travel between regions where different genotypes are more common can be exposed to different HCV genotypes, leading to a mixed infection. HCV is transmitted through contact with blood, such as through contaminated blood products or medical equipment, blood transfusions, kidney dialysis, or the sharing of drug injection equipment, such as syringes, or non-injection equipment, such as pipes, spoons, cotton balls, or straws for snorting drugs.
You May Like: Signs You Have Hepatitis B
Do Genotypes Change Over Time
A viruss genotype usually stays the same. Genetic changes, or mutations, can occur at random or in response to the environment. Some mutations are harmless, but others can affect how well a patient responds to treatment. New HCV treatments include more than one drug to prevent drug resistance from happening by targeting more than one step in the virus life cycle. However, if patients miss treatment doses, this can lead to genetic mutations, which cause resistance to HCV treatment .
HARDER TO TREAT GENOTYPE 3
Genotype 3 is the second most common HCV subtype in the world, particularly in Northern Europe, South Asia, and Southeast Asia. It can pose more difficult health problems for people with HCV, including more rapid progression of liver disease, increased rates of steatosis , and a higher risk for cancer . Genotype 3 has been associated with unique characteristics, such as how it creates resistance to insulin and how it causes the liver to break down fats, which make it harder to treat with DAAs.
People infected with genotype 3 are the most challenging to treat if they:
- have previously tried treatment
- have cirrhosis, and
- have , which is a life-threatening condition leading to liver failure.
Genotype 3 often requires longer treatment and does not achieve strong cure rates. There are lower cure rates in patients with cirrhosis.
Where Are The Different Hepatitis C Genotypes Found
HCV has evolved over time into 7 distinct genotypes , with at least 67 subtypes. Hepatitis C genotypes are distributed throughout the world, with some genotypes being predominant in certain geographic areas. HCV genotype 1 is the most common genotype in the US and Europe, accounting for 60% to 70% of HCV. HCV genotype 1a is more common than 1b in the US. 3
Hepatitis C genotypes 1 and 3 infection are associated with more aggressive liver disease, with increased risk for cirrhosis and fibrosis, as well as greater risk for hepatocellular carcinoma. After genotype 1, HCV genotype 2 is the next most common HCV genotype in the US, where it accounts for approximately 10% of cases. HCV genotypes 3-6 are more common in Africa, the Middle East, the Far East, India, and Australia. Genotype 7 has only been isolated in a limited number of cases. Genotype 3 is associated with increased risk for development of fatty liver disease . 1,4
Hepatitis C genotypes are substantially different from one another in their genetic make-up, which explains why each genotype responds differently to antiviral treatments.1,4
Also Check: Hepatitis B Vaccine Schedule For Baby
Retreatment Of Persons With Prior Peginterferon And Ribavirin Failure
The latest version of the AASLD-IDSA HCV Guidance no longer provides specific recommendations for retreatment of persons with a history of peginterferon plus ribavirin therapy, with or without an earlier generation direct-acting antiviral agent . The AASLD-IDSA HCV Guidance notes that these individuals respond to retreatment similar to treatment-naïve persons, thus implying the treatment approach should be the same as with treatment-naïve individuals. Although the pool of persons with a history of failure with a peginterferon-based regimen who need retreatment is small and diminishing, there are some individuals with this treatment history who need retreatment and may require special consideration that differs from that of treatment-naïve individuals. The following outlines a few of these key considerations based on available data and previous guidance that should be noted when retreating an individual with a history of prior treatment failure with peginterferon plus ribavirin, with or without an earlier generation DAA . Note that except for the 8-week option of glecaprevir-pibrentasvir , when retreating these individuals with first-line DAA combinations that have pangenotypic activity , the treatment will be the same as their treatment-naïve counterparts.
Treatments For Different Genotypes
DAAs are the most common treatment that directly targets the hepatitis C virus. A person may also need to take other medications, depending on the genotype of the virus and any complications of the infection.
The best treatment option tends to depend on whether or not the person has cirrhosis. The options below apply to people who have not previously received treatment for hepatitis C.
The following medications are recommended for genotypes 1a and 1b:
- elbasvir and grazoprevir
- ledipasvir and sofosbuvir
- glecaprevir and pibrentasvir
- sofosbuvir and velpatasvir
If a person has genotype 2 or 3, a healthcare provider may choose to prescribe glecaprevir and pibrentasvir or sofosbuvir and velpatasvir.
For people who have genotype 4, they may prescribe glecaprevir and pibrentasvir, sofosbuvir and velpatasvir, elbasvir and grazoprevir, or ledipasvir and sofosbuvir.
For people with genotypes 5 or 6, they may describe a combination of these medications. As one study paper concludes, developing more effective treatments and a better understanding of the prevalence of the condition will require more research.
The right treatment varies from person to person. When recommending a course of action, a healthcare provider will take into consideration previous treatments, medical history, overall health, and any complications of hepatitis C.
Don’t Miss: How Do You Know If You Have Alcoholic Hepatitis
Management Of Hepatitis C Genotype 1 Monoinfection
Makia Dove, PharmD CandidateMonika N. Daftary, PharmD, BCPS, AAHIVPProfessorCollege of Pharmacy, Howard UniversityWashington, DC
US Pharm. 2014 39:75-79.
ABSTRACT: Hepatitis C is a common cause of end-stage liver disease in the United States. Historically, standard treatment for hepatitis C virus genotype 1 was peginterferon alfa plus ribavirin. Treatment remained unchanged until 2011, when the direct-acting antivirals boceprevir and telaprevir were approved by the FDA. Standard treatment of HCV genotype 1 now consists of peginterferon alfa, ribavirin, and a DAA. The addition of a DAA to standard treatment helped shorten the duration of therapy, but the initially approved agents were not without adverse effects . In late 2013, two additional DAAssimeprevir and sofosbuvirwere approved by the FDA. Both of these agents feature improved dosing-frequency and AE profiles.
HCV is classified into six different genotypes , with each genotype divided into subtypes. HCV genotype 1, which is the most common cause of hepatitis C infection in the U.S., is the most difficult form to treat.4