Actions For This Page
- Recent advances in antiviral treatment have led to the development of new highly effective drugs for the treatment of all types of hepatitis C.
- The new hepatitis C treatments are sofosbuvir with ledipasvir sofosbuvir daclatasvir and ribavirin .
- These new treatments are now available on the Pharmaceuticals Benefits Scheme.
What Are Genotypes And Do They Matter
Six different genotypes of hepatitis C have been identified. Genotypes 1 and 3 are the most common causes of hepatitis C in Australia and make up 90 per cent of all cases. They are important because they help determine the treatment you need. Unlike in the past, however, your genotype is not important in terms of the chance of cure. With the treatment drugs, all six genotypes have a very high chance of cure.
Ombitasvir Paritaprevir And Ritonavir Tablets Co
This is a relatively new group of medicines that treat genotype 1 hepatitis.
Facts about the drug pack include:
- Treatment time is 12 or 24 weeks.
- Dosage is a pack of tablets containing 12.5 mg of ombitasvir, 75 mg of paritaprevir, and 50 mg ritonavir, taken once daily in the morning, and one 250 mg tablet of dasabuvir taken twice daily with a meal.
- Common side effects of this group of drugs include nausea, itching, and trouble sleeping. If the person also takes ribavirin, side effects include tiredness, nausea, fatigue, and skin reactions.
The following medications may be effective for genotype 2:
Don’t Miss: How To Cure Hepatitis C Naturally
What Do Experts Suggest About Diagnosis And Treatment Of Hcv
Both the United States Preventive Services Task Force and the Centers for Disease Control and Prevention recommend that all “baby boomers” — those born from 1945 to 1965 — be tested for HCV.
This group of adults represents about 75% of all cases, but most are not currently diagnosed. People at high risk for the hepatitis C virus includes those with a history of IV drug use and those who received blood transfusions before 1992, but many other risk factors exist.
Increased rates of screening and diagnosis will likely result in accelerated demand for treatments.
New oral combination treatments for HCV have been shown to result in HCV cures for most newly diagnosed patients.
What Are The Side Effects Of Treatment
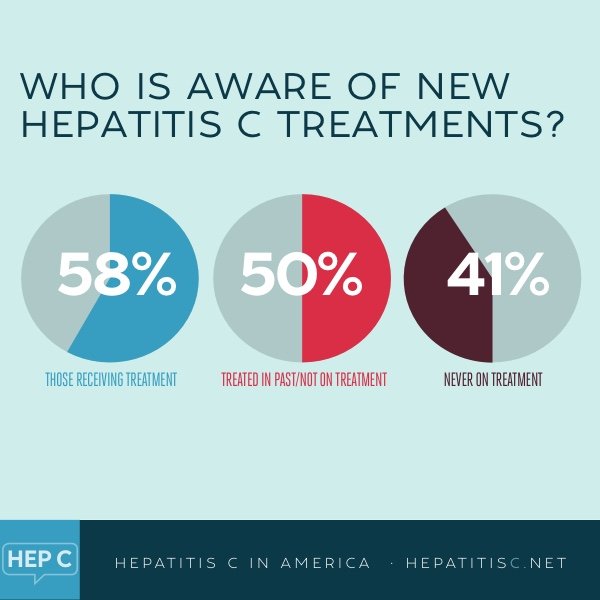
Some people stop therapy because of side effects. Since hepatitis C can lead to liver damage, cirrhosis, and liver cancer if not treated, its vital to stick with a treatment plan.
Newer drugs have fewer severe side effects than pegylated interferon and ribavirin. Nevertheless, you may feel some effects while taking hepatitis C medication. Side effects can include:
- nausea, vomiting, or diarrhea
- appetite loss or weight loss
Serious side effects can occur with pegylated interferon and ribavirin treatment. If youre taking these medications, you should be monitored for these serious side effects:
- anemia
- thrombocytopenia
- light sensitivity in the eyes
- trouble breathing because of lung tissue inflammation
- suicidal thoughts, depression, or irritability
- thyroid disease
- elevated liver enzymes
- autoimmune disease flares
Some medications arent recommended if theres evidence of liver damage, like cirrhosis . A co-infection with HIV also affects medication options.
Also Check: Hepatitis A Curable Or Not
Exacerbation Of Chronic Viral Hepatitis
In the treatment of chronic hepatitis B, HBe seroconversion was sometimes preceded by transient and moderate worsening of serum transaminases, but severe exacerbation of chronic hepatitis B infection and fatal liver failure can occur. Such fatalities were reported in under 0.5% of patients with hepatitis B . Patients with active cirrhosis or a previous history of decompensated cirrhosis are particularly susceptible to these complications .
Acute exacerbation of hepatitis is an extremely rare complication of chronic hepatitis C treatment. An exaggerated immune response to hepatitis virus was supposedly the cause of acute icteric hepatitis in two patients .
-
A 43-year-old man had a moderate rise in hepatic transaminase activities after 4 weeks of interferon alfa treatment. His liver tests normalized after withdrawal, but the aspartate transaminase activity increased dramatically shortly after treatment was restarted. His condition rapidly deteriorated, with a diagnosis of hepatorenal failure, and he finally required liver transplantation. Histological examination of the liver showed advanced micronodular cirrhosis, a feature not found on pretreatment liver biopsy.
In another study, only four of 11 241 patients treated with interferon alfa died of fulminant liver failure .
Talia B. Baker, Juan Carlos Caicedo, in, 2017
Getting Tested Is The Only Way To Know If You Have Hepatitis C
A blood test called a hepatitis C antibody test can tell if you have been infected with the hepatitis C viruseither recently or in the past. If you have a positive antibody test, another blood test is needed to tell if you are still infected or if you were infected in the past and cleared the virus on your own.
- Are 18 years of age and older
- Are pregnant
- Currently inject drugs
- Have ever injected drugs, even if it was just once or many years ago
- Have HIV
- Have abnormal liver tests or liver disease
- Are on hemodialysis
Read Also: How Can Hepatitis B Be Transmitted
History Made: Sovaldi And Harvoni
In October 25, 2013 the FDA Advisory Committee unanimously recommended approval of Gilead’s sofosbuvir for chronic HCV genotypes 1-4. The FDA announced final approval of Sovaldi on December 6, 2013. Historically, Gilead’s Sovaldi sales surpassed $10 billion in 2014, a record for any previous brand name launch at that time.
Harvoni contains sofosbuvir and the antiviral NS5A inhibitor ledipasvir. Harvoni — also a Gilead drug — hit mega-blockbuster status, too.
However, newer approvals have put pressure on sales of these original oral Hep C agents. Since the approval of Sovaldi and Harvoni, 6 additional oral HCV treatments have been approved.
Why The High Costs
At this time, theres a short list of blockbuster HCV drugs. Because the FDA only recently approved these drugs, the companies that manufacture them have market exclusivity. That means only these companies can promote and sell the drugs. It also means there are no generic versions of these drugs yet. Generics are typically much cheaper than brand-name versions.
The FDA determines how long this period of exclusivity will last. During this time, the pharmaceutical companies have a lot of freedom in establishing prices. And those who developed the new HCV drugs have set the pricing bar high.
The table below highlights the average cost of treatment for the combination DAAs currently available. Most of these drugs take at least 12 weeks to cure HCV, while the most recently approved drug, Mavyret, can take only eight weeks.
| Generic name | |
| $94,800 |
These costs are averages derived from information provided by www.goodrx.com. They were current at the time this article was published.
Also Check: How Long Does A Person Live With Hepatitis C
Where Can I Go If I Have Further Questions Or Need More Information
- Your local GP and pharmacist can provide you with more information on the new treatments, including if they are right for you. To find a GP, please click here
- The Victorian Government funds a range of community organisations to provide information, care and support to people living with hepatitis C, and on the new treatments. For more information, please visit:
- Hepatitis Victoria’s website or their Hepatitis Infoline on 1800 703 003or refer to the Hepatitis Victoria, PBS factsheets
Why Cure Hep C
Curing your hep C clears the virus from your body. It reduces liver inflammation and can help reverse fibrosis and even cirrhosis.
Live free from the worry of hep C knowing that you no longer have hep C can help you feel better about yourself. For example, you may no longer feel worried about passing hep C to other people. There has been no better time to think about hep C treatment.
Find out more about the benefits of clearing hep C call the Hepatitis Infoline.
|
Grace talks about her experience of being cured of hepatitis C with new, highly effective treatments. Theres never been a better time to be cured of hep C. |
Read Also: Hepatitis B Liver Cancer Treatment
Will Community Pharmacies Be Able To Dispense These New Hepatitis C Drugs
Community pharmacists will be able to dispense the drugs. However, because these are new drugs, it may take time for pharmacies to order in sufficient stock to meet demand.
This means that patients may need to wait a couple of days after providing their script for the drugs to be available from their local pharmacy.
Ifn Monotherapy In Acute Hepatitis C
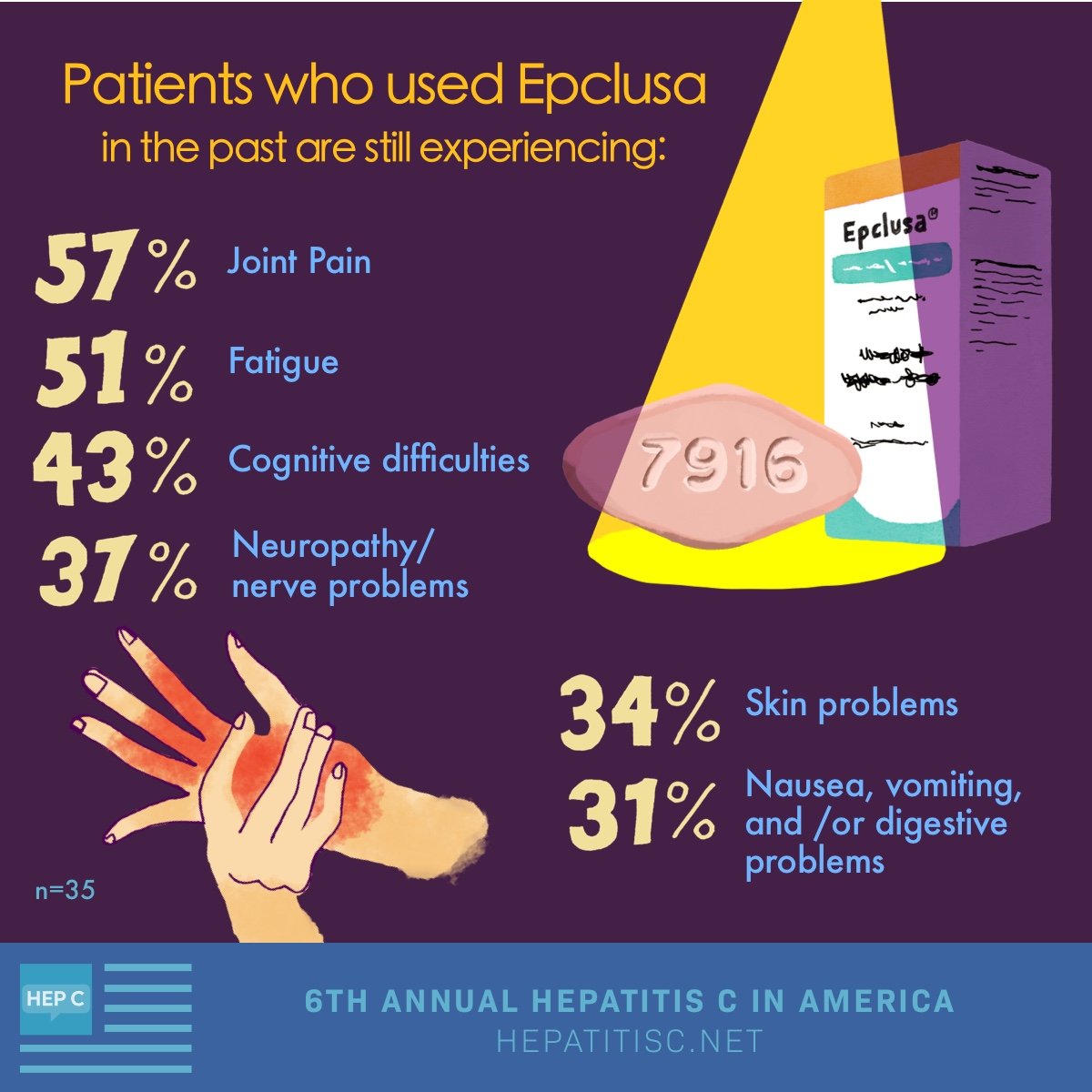
Although the short courses of standard IFN monotherapy introduced in the 1980s by Hoofnagle et al, Davis et al, and Di Bisceglie et al led to sustained improvement in liver disease and loss of virus in less than 10% of patients, these therapies were the first to cure chronic viral hepatitis.
Jaeckel et al reported that treatment with IFN alfa-2b prevented chronic infection in 98% of a group of 44 German patients with acute hepatitis C. In this study, patients received 5 million U/day of IFN alfa-2b subcutaneously for 4 weeks and then three times per week for another 20 weeks the IFN alfa-2b was well tolerated in all patients but one.
Because it has the poorest safety profile of all the HCV antiviral agents, with few exceptions PEG-IFN is no longer recommended in combination regimens. Spontaneous resolution of acute HCV infection may occur in 15% to 50% of patients. Monitoring for spontaneous clearance for a minimum of 6 months before initiating any treatment is therefore recommended.
References
World Health Organization. Hepatitis C: fact sheet. Available at . Updated: October 2017 Accessed: January 23, 2018.
Frank C, Mohamed MK, Strickland GT, et al. The role of parenteral antischistosomal therapy in the spread of hepatitis C virus in Egypt. Lancet. 2000 Mar 11. 355:887-91. .
Kim A. Hepatitis C virus. Ann Intern Med. 2016 Sep 6. 165 :ITC33-ITC48. .
Recommended Reading: Hepatitis B How Do You Get It
What’s Involved With An Hcv Diagnosis
Discuss your risk factors for hepatitis C infection with your healthcare provider. Drug abuse and having a blood transfusion prior to 1992 may have put you at risk, but there are other risks factors, too.
Hepatitis C screening starts with a blood test to look for viral antibodies. HCV antibodies can be detected in the blood within 2 to 3 months after infection. If this test is positive, a confirmatory blood test would be ordered.
An HCV viral load may be ordered to determine your chances for responding to treatment. In addition, HCV genotyping can help to guide the best treatment option and duration.
Your doctor may also order liver function tests – known as AST, ALT and GGT tests – to monitor the health of your liver.
A liver biopsy, usually performed as an outpatient surgical procedure, may be needed to determine the level of liver damage.
How Much Will It Cost Patients To Access The New Drugs
The Pharmaceutical Benefits Scheme listing means that hepatitis C patients will only pay the normal co-payment for the new drugs. The co-payment is currently worth $6.20 for patients with concessional healthcare cards and $38.30 for general patients without concessional healthcare cards, per drug, per month. For some patients needing three drugs, for example, the co-payment will therefore be $114.90 per month, or $18.60 per month concession.
More information on co-payment charges can be found on the Commonwealth Department of Healths website
Read Also: How Do You Know If You Have Hepatitis C
How Is Hepatitis C Treated
Hepatitis C is treated using antiviral drugs.
Treatment in the first 6 months focuses on:
- treating symptoms
- preventing the spread of the disease
- preventing complications, such as liver damage
For someone who has the disease beyond 6 months, treatment includes a combination of medications. However, not everyone with this form of hepatitis C will need treatment.
Whether or not you are getting treatment, you can help lower the risk of damage to your liver by:
- avoiding alcohol
What Will My Doctor Need To Know To Treat Me
If you want to be assessed for treatment, you need to make an appointment with a doctor. They will be mostly interested in the condition of your liver. Your doctor will organise, if possible, for you to have a Fibroscan examination. If Fibroscan is not available, your doctor will probably use an APRI test. This is an online calculator that estimates the health of your liver. It involves a blood test called a liver function test.
Dont forget, its very important to get a PCR test 12 weeks after finishing treatment this will mean the doctor can make sure you are cured.
You May Like: Which Is Worse Hepatitis B Or C
Can Hepatitis C Be Treated
Yes, since 2010 enormous progress has been made in the treatment of chronic hepatitis C. New therapies called direct-acting antivirals are pills that act on the virus itself to eradicate it from the body, unlike older medicines like interferon injections which work by stimulating an immune response. These new treatments are very effective and can achieve cure rates of over 90%. In most situations now, there is no need for interferon, which was responsible for many of the side effects previously associated with HCV treatment. The new treatment combinations require shorter treatment durations , have reduced side effects and appear to be effective at all stages of the disease.
Because these new therapies are very new, they remain very expensive. As such, drug coverage from both government and private companies may require that your liver disease has progressed to a certain stage before they are willing to cover the cost of these drugs.
Your primary care physician may refer you to a specialist to determine whether you are eligible for treatment. A specialist will help you decide which drug therapy is best for you based on the severity of your liver disease, your virus genotype and whether or not you have been treated in the past.
Nonstructural 5b Polymerase Inhibitors
The RdRp is vital to HCV replication, acting to catalyze RNA synthesis and genome replication. Nucleoside inhibitors arrest RNA synthesis, while nonnucleoside inhibitors bind and disrupt the RdRp function.
The nucleoside inhibitors are analogues that are incorporated into the viral RNA genome by the RdRp, causing termination of further replication, and competitively bind the active polymerase site. Single mutations can lead to resistance however, there is some evidence that mutations also seem to decrease viral fitness. This class of NS5B polymerase inhibitors has a high barrier to resistance and works broadly against genotypes with intermediate potency. Sofosbuvir was the first available NS5B nucleoside inhibitor .
Nonnucleoside inhibitors inhibit the RdRp by binding an allosteric site in a noncompetitive fashion, which changes the biochemical activity of the polymerase. They have a low barrier of resistance. Beclabuvir, an indole derivative, binds the thumb I subdomain on the RdRp with potent activity but has reduced activity against HCV genotypes 2 and 6 infections. Dasabuvir, a benzothiadiazine derivative, binds the palm I site on the RdRp, causing changes to the active site and preventing transcription., Deleobuvir and radalbuvir are additional nonnucleoside inhibitors .
Recommended Reading: How Can You Contract Hepatitis B
How Will My Provider Monitor Me During The Treatment
Your provider will meet with you during treatment to review how well you are tolerating treatment and review laboratory results. Laboratory tests help keep tabs on your health, track the viral load, and determine your response to treatment. You will be given specific dates to go get your blood tested at the lab during and after the treatment.
Use Of Oral Daas In Children With Hcv

The development of the oral direct-acting antivirals has transformed the treatment of adults and children with HCV. It is estimated that there are 23,000 to 46,000 children in the U.S. with HCV infection. The occurrence of HCV in children is much less frequent than in adults and is usually acquired at birth.
Treatments are available for children as young as 3 years of age. For children under 3 years of age, treatment should be deferred until at least the 3rd birthday, as many cases will resolve on their own.
- Approved for the treatment of chronic HCV in patients as young as 3 years of age, regardless of HCV genotype or liver disease severity. A new oral pellet formulation was approved in 2021 as well, for children who cannot swallow tablets. Dosing in children is based on weight, and in those less than 6 years old, give the pellets with food to improve palatability.
Read Also: How Do They Check For Hepatitis C
Treatment For Chronic Hepatitis C
Chronic Hepatitis is a long-term condition that can affect your liver. Antivirals will also be used to treat chronic conditions, but in some cases, a liver transplant may be recommended. During the surgery, the damaged parts of the liver will be removed and replaced with the healthy donor liver. After treatment, the virus may return, and antivirals may be again prescribed.
Will A Specialist Need To Be Involved
In order to prescribe, general practitioners including physicians with expertise in viral hepatitis, will be required to first consult with a gastroenterologist, hepatologist or infectious diseases physician to ensure patients with liver disease or other complex needs are appropriately referred to specialist care. A face to face consult with the specialist is not required and patients with complex needs will likely be referred to specialist care where appropriate.
Patients affected by hepatitis C with severe or advanced liver disease may still need to access the treatments under the care of a specialist – such as a gastroenterologist, hepatologist, or an infectious disease physician with experience in treating chronic hepatitis C infection.
Recommended Reading: Royal Canin Veterinary Diet Hepatic