The Dawn Of Interferon
The addition of DAAs to PEG-IFN plus RBV as part of triple therapy vastly improved outcomes for patients with CHC. Superior rates of SVR were reported, alongside shortened treatment durations for certain cohorts. However, triple therapy still involved interferon as a mainstay of treatment, bringing with it unpleasant side effects and weekly injections. These factors led to the development of new interferon-free regimens, combining various DAAs, with or without RBV. Interferon-free regimens vary depending on genotype and presence of cirrhosis, and are summarized in Table 2. Those interferon-free regimes currently licensed by the US Food and Drug Administration for use in the US are described in the review by Zhang et al.84
My Background In History
This is a brief history of the Hepatitis C virus. In 2014 G I was doing my Ph.D. at the University of Tasmania when I became very sick and discovered that I had Hepatitis C. The severity of my illness forced me to quit my Ph.D.
In 2015 I traveled to India and cured the Hepatitis C infection using generic Sovaldi. Since that time I have devoted my life to helping people with Hepatitis C access affordable treatment.
I have a Masters Degree in History and in this video, I briefly explain the history of Hepatitis C and Hepatitis C treatment. If you need support with your Hepatitis C please join my Hepatitis C
Case History Of Hepatitis C, Chronic History Of Hepatitis C, History Of Hep C, History Of Hep C Treatment, History Of Hep C Virus, History Of Hepatitis C Treatment, History Of Hepatitis Type C, Natural History Of Hepatitis C Infection, The History Of Hepatitis C, The History Of Hepatitis C Virus
- Post Categories
The Future Of Hepatitis C Therapy
With such high rates of success with current treatments, it may seem like the hepatitis C story is in its final chapters, but it is not over yet. A vaccine against hepatitis C would cause the prevalence of the disease to plummet, but efforts to produce a vaccine, while still under way, have not yet been successful. While hepatitis A and B have vaccines, the hepatitis C virus is more variable than either of these viruses, which, along with other factors, complicates vaccine development efforts. Additionally, the current drugs show great promise, but the costs of the more successful FDA-approved DAA treatments are extremely high, which present a significant obstacle to many with the disease. But the research has come a long way. From the early investigations into a mysterious new virus, to the identification of the culprit, and the rigorous work to develop an effective treatmentthe story of hepatitis C is definitely a thriller.
Don’t Miss: What Are The Symptoms Of Hepatitis B Virus
A Researcher Reflects On Progress Fighting Hepatitis C And A Path Forward
The hepatitis C virus was discovered in 1989 research thats now earned a Nobel Prize.
When I began my medical career in Hong Kong in the early 1980s, I chose to focus on hepatitis B, in part because it was very common and because the hepatitis C virus had not yet been discovered. I witnessed the devastation that this virus caused cirrhosis, liver failure and liver cancer and the lack of treatments we could offer to patients.
Back then, scientists knew there was another type of hepatitis, but no one could identify it, so we called it non-A, non-B hepatitis. I would never have imagined that during the course of my career I would witness the discovery of what came to be known as hep C and the development of a cure for nearly all patients with chronic hepatitis C in 2014.
Underscoring the importance of these discoveries for global human health, this years Nobel Prize in Physiology or Medicine was awarded jointly to Harvey J. Alter, Michael Houghton and Charles M. Rice for the discovery of the hepatitis C virus.
Effective treatment for hepatitis C has become even more relevant today in light of the recent surge in new cases of hepatitis C due to rising opioid use.
How Do Doctors Treat Hepatitis C
Doctors treat hepatitis C with antiviral medicines that attack the virus and can cure the disease in most cases.
Several newer medicines, called direct-acting antiviral medicines, have been approved to treat hepatitis C since 2013. Studies show that these medicines can cure chronic hepatitis C in most people with this disease. These medicines can also cure acute hepatitis C. In some cases, doctors recommend waiting to see if an acute infection becomes chronic before starting treatment.
Your doctor may prescribe one or more of these newer, direct-acting antiviral medicines to treat hepatitis C:
You may need to take medicines for 8 to 24 weeks to cure hepatitis C. Your doctor will prescribe medicines and recommend a length of treatment based on
- which hepatitis C genotype you have
- how much liver damage you have
- whether you have been treated for hepatitis C in the past
Your doctor may order blood tests during and after your treatment. Blood tests can show whether the treatment is working. Hepatitis C medicines cure the infection in most people who complete treatment.
Hepatitis C medicines may cause side effects. Talk with your doctor about the side effects of treatment. Check with your doctor before taking any other prescription or over-the-counter medicines.
For safety reasons, talk with your doctor before using dietary supplements, such as vitamins, or any complementary or alternative medicines or medical practices.
Don’t Miss: Hepatitis B Core Ab Total
Exacerbation Of Chronic Viral Hepatitis
In the treatment of chronic hepatitis B, HBe seroconversion was sometimes preceded by transient and moderate worsening of serum transaminases, but severe exacerbation of chronic hepatitis B infection and fatal liver failure can occur. Such fatalities were reported in under 0.5% of patients with hepatitis B . Patients with active cirrhosis or a previous history of decompensated cirrhosis are particularly susceptible to these complications .
Acute exacerbation of hepatitis is an extremely rare complication of chronic hepatitis C treatment. An exaggerated immune response to hepatitis virus was supposedly the cause of acute icteric hepatitis in two patients .
-
A 43-year-old man had a moderate rise in hepatic transaminase activities after 4 weeks of interferon alfa treatment. His liver tests normalized after withdrawal, but the aspartate transaminase activity increased dramatically shortly after treatment was restarted. His condition rapidly deteriorated, with a diagnosis of hepatorenal failure, and he finally required liver transplantation. Histological examination of the liver showed advanced micronodular cirrhosis, a feature not found on pretreatment liver biopsy.
In another study, only four of 11 241 patients treated with interferon alfa died of fulminant liver failure .
Talia B. Baker, Juan Carlos Caicedo, in, 2017
Evolution Of Hcv Therapy
The ultimate goal of hepatitis C treatment is to reduce the occurrence of end-stage liver disease and its complications, including decompensated cirrhosis, liver transplantation, and HCC. Treatment success is assessed by sustained virologic response , defined by the presence of undetectable HCV RNA in blood several months after completing a course of treatment .
Figure 6.1. Evolution of chronic HCV therapies. DAA, direct-acting antiviral agent HCV, hepatitis C virus IFN, interferon PEG, pegylated RBV, ribavirin SVR, sustained virologic response.
Graham A.W. Rook, … Christopher A. Lowry, in, 2018
Recommended Reading: Patient Has Immunity To Hepatitis B Virus
Zeroing In On The Hepatitis C Virus
The era of direct-acting antivirals that specifically target HCV began in 2011 with the U.S. Food and Drug Administration approval of the first protease inhibitors. These drugstelaprevir and boceprevir, along with several similar drugs approved latertargeted the HCV protease that is critical for viral replication. When used in conjunction with peginterferon and ribavirin, protease inhibitors yielded SVR rates of up to 75 percent. However, this triple therapy was accompanied by additional side effects to those already present with peginterferon and ribavirin. Nevertheless, the success of HCV-specific protease inhibitors showed that the virus had vulnerabilities that could be exploited by a well-designed and properly administered drug.
More new anti-HCV drugs were developed and tested over the next several years. These new drugs included sofosbuvir and dasabuvir, which interfered with the activity of the HCV polymerase, an enzyme that is responsible for the viral replication. Members of a second class of drugs, ledipasvir and daclatasvir, targeted the NS5A region of the virus, which makes a structural protein critical for viral replication. Many of these drugs were initially tested in conjunction with peginterferon and ribavirin, or in combination with a protease inhibitor. Generally, the results were SVR rates of at least 80 percent.
About The Hepatitis C Virus
Hepatitis C is a blood-borne virus that predominantly infects the cells of the liver. This can result in inflammation and significant damage to the liver. It can also affect the livers ability to perform its essential functions. Although it has always been regarded as a liver disease – hepatitis means inflammation of the liver – recent research has shown that the hepatitis C virus affects a number of other areas of the body. These can include the digestive system, the lymphatic system, the immune system and the brain.
Hepatitis C was first discovered in the 1980s when it became apparent that there was a new virus causing liver damage. Before being properly identified in 1989 it was originally known as non-A non-B hepatitis. In 1991 a screening process was developed making it possible to detect HCV in blood samples. As a relatively new disease there are still many aspects of hepatitis C which are yet to be fully understood.
There are an estimated 150 million people worldwide chronically infected with hepatitis C. The level of infection, known as prevalence, varies widely from country to country. In some countries, such as Egypt, it is as high as15%. In the United States it is believed to be 1% and in the UK it is believed to be around 0.5%. The virus can only be transmitted by infected blood.
You May Like: Hepatitis C Screening Guidelines Cdc
Spontaneous Clearance Versus Chronic Infection
Most persons who acquire hepatitis C virus will develop chronic infection. Following acute infection, HCV is very successful in establishing persistent infection by evading the immune system. Although the mechanism for the high rate of viral persistence is not completely understood, several viral and host factors likely play a significant role. The actual rate of chronicity following initial infection with HCV is not well established in prospective studies, primarily because of the high percentage of persons who have asymptomatic or unrecognized early infection. The chronicity rate has been estimated from cross-sectional population-based studies, such as the National Health and Nutrition Examination Survey , and numerous retrospective studies. Overall, an estimated 55 to 85% of persons who become infected with HCV will develop chronic HCV infection.
Global Distribution Of Hepatitis C Genotypes
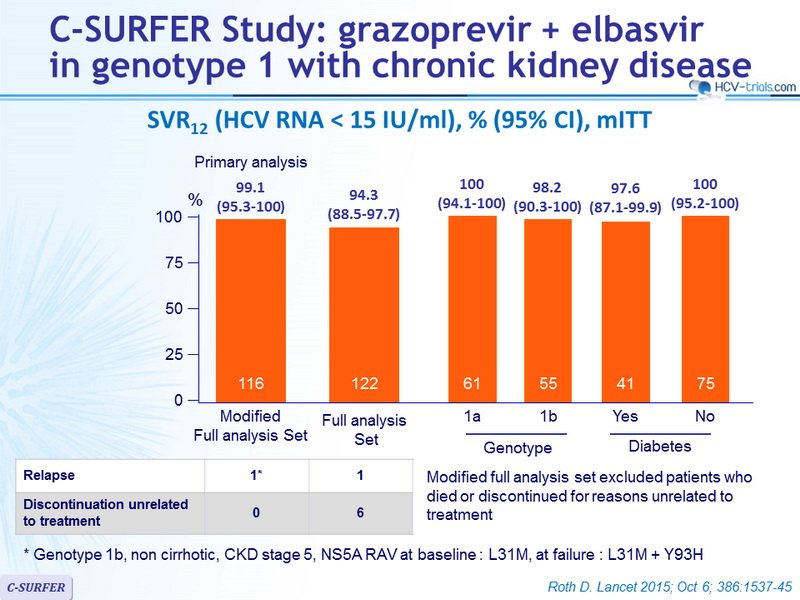
HCV follows a worldwide distribution, with Africa and Central and East Asia being most affected.8 The most common genotype both worldwide and in Europe and North America is GT1, accounting for 49.1% of CHC cases.1 GT1 infection can be further subdivided into two major classes: 1a and 1b.3 While GT1a accounts for the majority of CHC GT1 cases in North America, the majority of CHC GT1 cases worldwide are due to GT1b 5 . GT3 is the second most common genotype globally, accounting for 17.9% of CHC cases. Worldwide, GT4, GT2, and GT5 account for 16.8%, 11%, and 2% of cases, respectively.1 According to recent estimates, GT6 infection is the least common, accounting for 1.4% of CHC cases.1 Genotype distributions in Europe follow a similar pattern, with GT1 and GT3 accounting for the majority of CHC cases .9 Globally, the majority of GT2 and GT6 cases are found in East Asia. GT4 is most commonly found in North Africa and the Middle East, particularly in Egypt following the anti-schistosomal treatment program that left many millions infected with HCV.5,10 GT5 is primarily found in South Africa.5
Distribution of GT1a versus GT1b. Reproduced from Messina JP, Humphreys I, Flaxman A, et al. Global distribution and prevalence of hepatitis C virus genotypes. Hepatology. 2015 61:7787. Creative Commons license and disclaimer available from: .5
Abbreviations: GT, genotype HCV, hepatitis C virus.
Don’t Miss: Is Hepatitis The Same As Hiv
Progression Based On Disease Severity
Two groups that represent opposite ends of the disease spectrum progress at differing rates, namely those who are immune suppressed and those who are HCV infected but who have persistently normal aminotransferase levels.
Immunocompromised individuals
Hepatitis C virus-infected persons who are immunocompromised, either because of HIV co-infection or through immunosuppressive treatments , have a higher rate of progression to a more serious outcome than do those who are mono-infected. Relative to immunocompetent infected persons, immune suppressed persons have an approximately two-fold increase in the frequency and rate of development of cirrhosis.
Opioid Epidemic Homeless Lead To Rise In Hepatitis B And C Infections
In the United States, the number of new hepatitis B virus and hepatitis C virus infections has been decreasing for many years, but this trend has been reversed during recent years due to the opioid epidemic as more people use injection drugs, share needles or other paraphernalia and practice high-risk sexual behavior. This is particularly true for hepatitis C, where the number of new cases in the past 10 years has more than doubled, highlighting the need for a preventive vaccine, which is a vital tool to eliminate hepatitis C. The increase in number of new cases of hepatitis B is smaller and mainly seen in adults in their 30s because most younger persons have benefited from hepatitis B virus vaccination.
When we talk about viral hepatitis, the focus is on hepatitis B and C because they can cause chronic infection, while hepatitis A causes only acute infection and will not lead to cirrhosis or liver cancer. However, since 2016, many states in the U.S. have witnessed outbreaks of hepatitis A. The Centers for Disease Control and Prevention received more than 2,500 reports of hepatitis A between January 2017 and April 2018 associated with person-to-person transmission, with risk factors in two-thirds of these cases being drug use or homelessness or both. In Michigan, where I live, 859 cases of hepatitis A, including 27 deaths, were reported between July 2016 and June 2018. We can prevent hepatitis A through vaccination and improved hygienic conditions.
Also Check: How Long Does A Person Live With Hepatitis C
Cost Of Hepatitis C Medicines
The newer direct-acting antiviral medicines for hepatitis C can be costly. Most government and private health insurance prescription drug plans provide some coverage for these medicines. Talk with your doctor about your health insurance coverage for hepatitis C medicines.
Drug companies, nonprofit organizations, and some states offer programs that can help pay for hepatitis C medicines. If you need help paying for medicines, talk with your doctor. Learn more about financial help for hepatitis C medicines.
A Pricey Drug And New Generics
The first combo pill with two drugs that inhibits different steps in hepatitis C replication was approved by the FDA in 2014. This pill is taken once a day for 8-12 weeks, has little to no side effects and improved the cure rate to 90-95%. It was hailed as a magical cure, but it came with a price tag of US$94,500 for a 12-week course of treatment. That led many insurers in the United States and national health departments in other countries to limit access to treatment.
Since then, several othercombo pills withsimilar cure rates that are equally well-tolerated have become available, and the cost has markedly decreased. In addition, low-cost generics and special pricing arrangements are available in many resource-limited countries.
While the current price of hepatitis C virus drugs is still very high, one needs to remember that for 95 percent of patients, this is a cure. It is unlike medicines for many illnesses that need to be taken for a long time, sometimes for the rest of the patients lives. Indeed, a cure for hepatitis C virus has allowed some patients who were on the liver transplant waiting list to reverse their liver failure, making transplantation unnecessary. This is good news not only for these patients but also for others on the waiting list.
Read Also: How To Tell If You Have Hepatitis C
Innovating Against A Silent Killer
Imagine taking an injection and a pill that made you feel every day worse than you ever felt from the infection that was being treated. Alexea Gaffney-Adams, MD, infectious disease specialist
Hepatitis C is dubbed the silent killer as it typically progresses without symptoms, often leaving patients unaware they are infected until their condition is very serious. HCV damages the liver slowly over many years, often moving from inflammation to scarring to permanent, irreversible scarring .
Once a patient has cirrhosis, the liver is unable to heal itself, and this condition can rarely be reversed. For those with end-stage liver disease, treatment is more focused on preventing further damage in an effort to avoid complications, including liver cancer, liver transplantation, and premature death. Hepatitis C has also been associated with other serious conditions, including diabetes, kidney disease, and depression.
In 1987, scientists working at Chiron Corporation, later acquired by Novartis, partnered with the CDC. Using a novel molecular cloning approach, they officially identified and named the virus hepatitis C in 1989.
Interferon was the first, and for a time, the only treatment for hepatitis C. An interferon is a protein produced by the bodys immune system in response to an infection. Side effects were debilitating, and many patients dropped out of what was a very long course of treatment. It was also a largely ineffective treatment.
The Hepatitis C Virus
Hepatitis is a general term for inflammation of the liver, which may be caused by viruses, toxins, drugs, or other factors. HCV was first identified in 1989 formerly it was known as non-A, non-B hepatitis. HCV is a member of the Flaviviridae family and is unrelated to other known hepatitis viruses. HCV must enter liver cells to carry out its life cycle once inside, the virus commandeers the cell’s ribosomes to reproduce, eventually killing the host cell.
There are at least six genotypes and numerous subtypes of HCV. Multiple quasispecies may co-exist in a single individual. Different subtypes have different geographical distributions and are associated with different rates of disease progression, severity, and response to treatment. Subtypes 1a and 1b together account for about three-quarters of HCV infections in the U.S.
Until recently, HCV could not be grown in the laboratory, making the study and testing of anti-HCV drugs difficult. This past summer, however, Ralf Bartenschlager and colleagues from the University of Mainz, Germany, reported they had devised a DNA mirror-image “replicon” of HCV’s genetic material that could be inserted into human cells to create a laboratory model of HCV infection.
Don’t Miss: Diagnostic Test For Hepatitis B