Infants Born To Hbsag
The routine schedule for these infants is a birth dose of monovalent HepB plus HBIG, then three routine doses of DTaP-IPV-HepB/Hib at ages 6 weeks, 3 months and 5 months.
All pregnant women should receive antenatal screening for hepatitis B infection by testing for HBsAg. Infants of HBsAg-positive mothers are to be notified at birth using the form HE1446: Consent for hepatitis B vaccine and hepatitis B immunoglobulin and notification to the Medical Officer of Health, available from the Health Ed website or the local authorised health education resource provider or public health unit.
Infants born to HBsAg-positive mothers should receive:
- 100110 IU HBIG neonatal, at or as close as possible to birth
- a birth dose of HepB which should be given at or as close as possible to birth .
If HBIG and/or HepB is inadvertently omitted, administer as soon as the omission is recognised. HBIG can be administered up to seven days post-delivery. If there is a delay for longer than seven days, seek specialist advice.
These infants should then continue as per the Schedule at ages 6 weeks, 3 months and 5 months. Serological testing is required at 9 months of age .
The vitamin K injection may also be given at the same time, in the same limb as the HBIG, but not at the same site.
Infants born to mothers who received oral antiviral therapy for chronic HBV must still receive the recommended neonatal HBIG/vaccine schedule. All other vaccines are administered as per the Schedule.
The Hepatitis B Vaccine And Immunosuppressants
If you are taking or about to start taking a medication that suppresses your immune response, let your healthcare provider know. Immunosuppressants may make certain vaccines less effective. Your healthcare provider may recommend that you get the hepatitis B vaccine at a particular time during your course of medication.
Dose And Administration Of Hepatitis B Vaccine
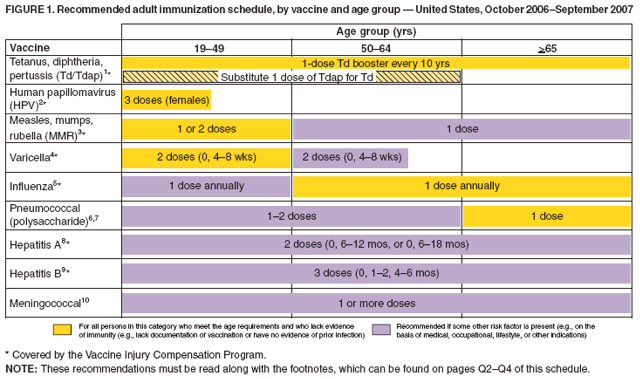
The dose for Engerix-B® and Recombivax HB® is 0.5 mL IM up to age 20 years or 1 mL IM for adults . The dose for Heplisav-B® is 0.5 mL IM for adults 18 years.
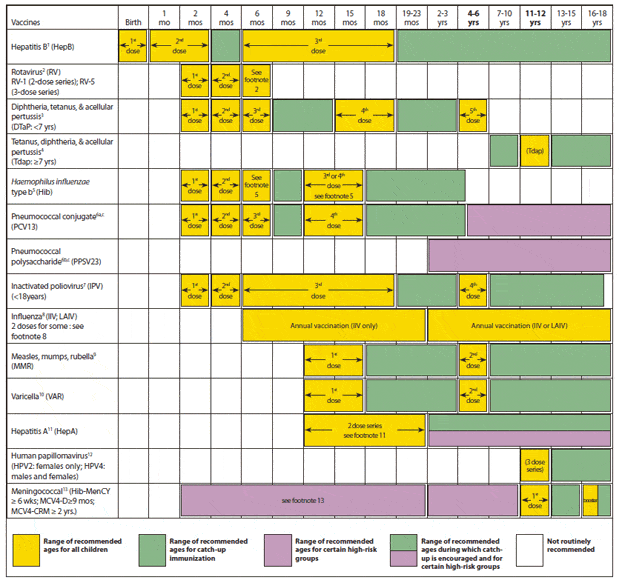
The vaccine is typically given to children in a 3-dose series at age 0 months, at 1 to 2 months, and at 6 to 18 months.
Infants who did not receive a dose a birth should begin the series as soon as feasible.
All children not previously vaccinated with HepB vaccine should be vaccinated at age 11 or 12 years. A 3-dose schedule is used the 1st and 2nd doses are separated by 4 weeks, and the 3rd dose is given 4 to 6 months after the 2nd dose. However, a 2-dose schedule using Recombivax HB® can be used the 2nd dose is given 4 to 6 months after the first.
The usual schedule for adults using Engerix-B® or Recombivax HB® is a 3-dose series with 2 doses separated by 4 weeks, and a 3rd dose 4 to 6 months after the 2nd dose. Heplisav-B® is given in 2 doses at least 4 weeks apart and can be given as a substitute in a 3-dose series with a different HepB vaccine. Heplisav-B® should not be given during pregnancy because safety data are not available on its use during pregnancy.
Unvaccinated adults who are being treated with hemodialysis or who are immunocompromised should be given 1 dose of Recombivax HB® 40 mcg/mL in a 3-dose schedule at 0, 1, and 6 months or 2 doses of Engerix-B® 20 mcg/mL given simultaneously in a 4-dose schedule at 0, 1, 2, and 6 months.
Don’t Miss: Hepatitis B Symptoms In Females
Interchangeability And Dosing Schedule
- 2-dose HepB vaccine series only applies when both doses consist of HepB-CpG, administered at least 4 weeks apart.
- Series consisting of a combination of 1 dose of HepB-CpG and a vaccine from a different manufacturer should do the following:
- Adhere to the 3-dose schedule minimum intervals of 4 weeks between dose 1 and 2, 8 weeks between dose 2 and 3, and 16 weeks between dose 1 and 3. However, if HepB-CpG is substituted for dose 2 of HepB-alum, a provider has the option of administering the next dose of HepB-CpG a minimum of 4 weeks from the previous dose for a complete series.
How Is Hepatitis B Treated
Immunoglobulin, a passive immunisation, can be administered within 12 hours of an at-risk exposure to help prevent HBV infection. Treatment for chronic disease is through the use of antiviral medications and medications containing naturally-occurring proteins. A liver transplant may be necessary in the event of severe liver damage.
A Hepatitis B vaccination schedule is the best way to prevent infection.
Read Also: If You Have Hepatitis B Can It Go Away
Eligible Adults Aged 18 Years And Older
|
0, 1 and 6 months |
Adult dialysis or adult liver or kidney transplant patients
These adults may have a reduced response to HepB, so three higher doses are recommended and funded.
See section 9.5.7 for information about post-vaccination serology.
Adult HIV patients
Adult HIV patients should receive four doses of HepB at 0, 1, 2 and 12 months.
Other eligible adults
The optimal dosing regime is three doses of 20 µg HepB given at 0, 1 and 6 months. See the manufacturers data sheet for sub-optimal accelerated HepB schedules if dosing is time constrained. For other eligible adults, see Table 4.8, Other special groups in section 4.6.
What Are The Symptoms
Following transmission and the average incubation period of 10 weeks , some of those infected may have no symptoms this is more common in children under 5 years of age. Between 30% and 50% of infected people older than 5 will experience gradually increasing fatigue, loss of appetite, nausea and pain in the right upper abdomen. A rash may present as one of the symptoms and also pain in the joints. These are followed by yellow discolouration of the skin and darkening of the urine. In a small number of cases, HBV infection results in rapid liver failure and death.
Up to 10% of adults and 30% of children who contract HBV will become chronic carriers this means they will recover but will always remain capable of transmitting the disease. Most babies infected at birth will develop chronic Hepatitis B infections and, with it, a higher risk of liver cancer or failure later in life.
You May Like: Is Milk Thistle Good For Hepatitis C
Facts About Hepatitis B
- Two billion people, or one in three, have been infected with hepatitis B worldwide. Of these, almost 300 million live with chronic hepatitis B. This means about 1 of every 26 people throughout the world are living with a chronic hepatitis B infection.
- Each year about 900,000 people die from hepatitis B worldwide, and about 2,000 of these deaths occur in the United States.
- Hepatitis B is transmitted through blood and is 100 times more infectious than HIV. An estimated one billion infectious viruses are in one-fifth of a teaspoon of blood of an infected person, so exposure to even a very small amount, such as on a shared toothbrush, can cause infection.
- Hepatitis B is sometimes referred to as the silent epidemic because most people who are infected do not experience any symptoms.
- Liver cancer accounted for about 5% of cancer deaths in the U.S. during 2020.
- Almost half of liver cancers are caused by chronic infection with hepatitis B.
- The World Health Organization recommends the inclusion of hepatitis B vaccine in immunization programs of all countries in 2019, more than 8 of 10 infants born throughout the world received three doses of hepatitis B vaccine.
Other Reported Adverse Events And Conditions
While serious events and chronic illnesses such as chronic fatigue syndrome, multiple sclerosis, Guillain-Barré syndrome, rheumatoid arthritis and sudden infant death syndrome have been alleged or reported following HB vaccination, no evidence of a causal association has been demonstrated in a number of studies.
Recommended Reading: What Is Hepatitis C Antibody Test
Infants Born To Mothers Who Have Hepatitis B: Hepatitis B Vaccine Schedules
*Please note that the first dose should be given as soon as possible. Additional doses require minimum time intervals between doses in order for the vaccine to be effective.
Protecting Your Baby
Infants born to women with hepatitis B must receive accurate doses of hepatitis B vaccine and hepatitis B immune globulin to ensure complete protection. In order to protect these infants, medications should be given immediately after birth in the delivery room or within the first 12-24 hours of life*.
* See Testing and Treatment During Pregnancy section for details. Please note that testing of all pregnant women for hepatitis B is a global recommendation.
3-Dose Vaccine Series for Infants
The World Health Organization recommends that infants born to hepatitis B positive mothers receive the first dose of the hepatitis B vaccine within 24 hours of birth, and ideally a dose of hepatitis B immunoglobulin . These shots must be followed by the additional vaccine doses given on the recommended schedule. In the U.S., infants should follow a 1 month and 6-month schedule for the additional two doses.
4-Dose Combination Vaccine Series for Infants
How Common Is Hepatitis B
One U.S. study following trends in hepatitis B infection over a three-year periodfound that 4.3% of the population had a past or present HBV infection.
Estimates suggest that about 240 million people around the world have chronic hepatitis B. Up to 1.89 million people in the United States have a chronic HBV infection.
Read Also: Signs Of Hepatitis C In Adults
What Is Hepatitis B
Hepatitis B is a highly contagious liver infection caused by the hepatitis B virus . The infection can range in severity from mild to acute. It may last just a few weeks or become a serious, chronic, and potentially fatal health condition.
The best way to prevent this infection is to get the hepatitis B vaccine. Heres what you need to know.
Global Burden Of Disease
Approximately two billion people worldwide had been exposed to HBV in 1995. In 2015, based on serological data, around 3.5 percent of the general population globally were infected with HBV and more than 250 million people were estimated to have chronic infection and these people remain at risk of developing cirrhosis and hepatocellular carcinoma. More than 90 percent of individuals with chronic HBV resided in the AsiaPacific region, where most countries have high prevalence rates of HBV infection and more than 99 percent of HBV-infected people in this region acquired infection through vertical transmission from their mother or in early childhood. As an example of this risk, 22.8 million out of 80 million people living in China with chronic HBV infection are women of child-bearing age. Acquisition of HBV during adulthood is associated with a high rate of symptomatic hepatitis but a low rate of chronic infection.
Also Check: Is Hepatitis A Vaccine A Live Virus
What Are The Uses For Hepatitis B Vaccine
Hepatitis B vaccine is used to prevent hepatitis B, a serious infection that affects the liver.
Most children are given their first shot at birth, followed by a 2nd shot at 1-2 months of age, and a 3rd shot at 6-18 months of age. Also, anyone who is 18 years of age or younger and hasnât received the vaccine should be vaccinated.
Additionally, all unvaccinated adults at risk for hepatitis B infection should be vaccinated. This includes:
- Partners or people infected with hepatitis B
- Men who have sex with men
- People who inject street drugs
- People with more than one sex partner
- People with chronic liver or kidney disease
- People under the age of 60 who have type 1 or 2 diabetes
- People with jobs that expose them to human blood or other body fluids
- People who live with a family member infected with hepatitis B
Immunogenicity And Effectiveness/efficacy Of Accelerated Vaccination Schedules In Different Populations
Table 1Overview of immunogenicity results according to vaccination schedule, in different populations
| Ref. | |
|---|---|
| Merck recombinant hepB | 0 1 6 |
| Merck recombinant hepB | 0 1 2 |
| Merck recombinant hepB | 0 1 12 |
| GenHevac B | 0 1 2 12 |
| GenHevac B | 0 1/3 3/4 12 |
| GenHevac B | 0 1 2 |
| GenHevac B | 0 1/3 3/4 |
| Bevac | 0 1 2 |
| EngerixB | 0 1 2 |
| RecombivaxHB | 0 6 |
| RecombivaxHB | 0 6 |
| RecombivaxHB | 0 6 |
| RecombivaxHB | 0 6 |
*Bold values refer to a higher antigen dosage schedule expressed in months 1/4 1/3 1/2 3/4 3/4 therefore correspond to 7, 10, 14, 21days, and 1.5months , respectively type of vaccination schedule: coded as S , SS , A , SA or P parentheses indicate schedules without the final dose § M/F: male/female ratio NA: not applicable ¶numbers and percentages either reported in the paper or calculated from the reported values.
Healthy adolescents
Few studies have examined the use of accelerated hepatitis B vaccination schedules in adolescents. Nevertheless, since the immunogenicity of many vaccines, including hepatitis B vaccines, is higher in younger persons, the results of studies in healthy adults can safely be extrapolated to younger age groups.4
Healthy adults
Travellers to areas of intermediate or high endemicity
Drug users
Lugoboni et al found a similar immunogenicity as compared with the general population, using the combined vaccine according to a standard vaccination schedule against hepatitis A and B in a population of drug users.37
Also Check: Hepatitis B Vaccine Dosage For Newborns
Compliance With Accelerated Vs Standard Vaccination Schedules In Different Populations
Table 2Overview of hepatitis B vaccine uptake according to vaccination schedule, in different atrisk populations
| Ref. | |
|---|---|
| Prisoners with intravenous drug use | 0 1 6 7 |
| Prisoners with intravenous drug use | 0 1/4 3/4 7 |
| Prisoners with intravenous drug use | 0 1/4 3/4 7 |
| MSM, IVDU, CSW and STI | 0 1 6 |
*Schedule expressed in months 0 1/4 3/4 therefore corresponds to 0.7.21days type of vaccination schedule: coded as S , SS , A or SA parentheses indicate schedules without the final dose numbers and percentages either reported in the paper, or calculated from the reported values
SW/MSM/Multiple partners/STI clinic attendants
Several studies have reported being able to administer three doses of hepatitis B vaccine to a higher proportion of the population targeted, when an accelerated or a superaccelerated schedule was used, at least the primary part of it. Unfortunately, few of these studies report immunogenicity data this is mainly due to the difficulties to administer three vaccine doses, and thus the low proportion that can actually be tested afterwards.39,40,41,42,43,44,45,46
A recent paper that did report immunogenicity data studied a shortened standard schedule as an alternative option, in a setting where other strategies are used to improve the compliance. Even if the 0.1.4months schedule failed to significantly improve the compliance, it offered equal protection within a shorter interval.47
Drug users
I Am A Healthcare Worker Who Did Not Develop Hepatitis B Antibodies After Immunization What Should I Do
Two versions of hepatitis B vaccine are available. One, called Heplisav-B, contains a novel adjuvant that was not present in previous versions used by adults . Some people did not respond to the older version hepatitis B vaccine. In fact, in a group of adults younger than 40 years of age who received two doses of the older version vaccine 75 of 100 were protected. Following the third dose, this number increased to 90 of 100. However, people older than 40 years of age were less likely to respond to the vaccine with increasing age. On the other hand, 90 to 100 of 100 adults 18 years of age and older respond to Heplisav-B, which was approved for use in 2018.
About 5-10 of every 100 children and adults younger than 40 years of age do not respond to the third dose of the hepatitis B vaccine. Some of these people will be recommended to get vaccinated again. About 5 of 100 people will still not respond after getting all recommended doses of both series. Note that children younger than 18 years of age cannot get Heplisav-B.
If the people who do not respond to vaccination are determined not to have chronic hepatitis B, they will be reliant on taking precautions to reduce the chance of exposure and relying on those around them for protection. In other words, these people will be reliant on herd immunity.
Recommended Reading: How Do You Get Acute Hepatitis
Reducing Hbv Infections And Deaths By 2030
As part of their plan to address HBV, the CDCs Division of Viral Hepatitis announced a goal to reduce HBV infections by 90% and HBV-related deaths by 65% in 2030.
The Department of Health and Human Services also announced a goal to eliminate viral hepatitis as a public health threat in the United States by 2030.
To help meet these goals, the CDC has outlined new hepatitis B vaccination recommendations for 2022. Vaccines can help prevent the disease and have reduced the number of reported hepatitis B cases by 90% since being introduced in 1982.
A hepatitis B vaccine is now recommended for adults ages 19 to 59 years without risk factor screening and disclosure. This helps increase vaccination coverage and decrease cases.
The vaccine is also recommended for adults ages 60 years or older with risk factors for hepatitis B. Risk factors can include having sex without a condom or barrier method, sharing needles, and having a job that exposes you to human blood, among others.
Its also recommended that adults 60 years or older without known risk factors get a vaccine. New clinical guidance advises providers to offer vaccinations to this group. Previously, clinical guidance instructed providers to administer the hepatitis B vaccine only when it was requested by a patient.
The CDC also continues to recommend hepatitis B vaccination for all infants and unvaccinated children under the age of 19 years.