Outcomes Of Hai Chemotherapy For Unresectable Disease
Conversion to resection is the goal of HAI and systemic chemotherapy, as resection is independently associated with prolonged OS . However, resectability of liver metastasis is surgeon and institution dependent, and therefore, reports of conversion to resection rates in available studies are difficult to interpret.
Available treatment options for unresectable CRLM limited to the liver were initially limited to systemic chemotherapy, with low conversion rates and poor OS. The first systemic chemotherapy used for metastatic CRC was systemic 5-FU, with a response rate of only about 20%, and average survival of approximately 11 months . Irinotecan and oxaliplatin were later developed and had a higher response rate and a slightly longer median survival of 1519.5 months . In a single institution study from 1988 to 1999, the conversion rate with chemotherapy was 12.5% . In more recent studies using modern combination therapy and targeted therapy, response rates of up to 81% and conversion rates up to 60% are reported with combination FOLFOXIRI and bevacizumab . Modern systemic chemotherapy has increased survival in metastatic CRC patients to 1830 months however, disease progression and eventual death secondary to liver failure remain the biggest clinical challenges. Thus, there is a continued interest in HAI and other locoregional therapies including radiofrequency ablation, stereotactic body radiation, and chemoembolization as methods to stave off hepatic disease progression .
Hai For Unresectable Bile Ducts Carcinoma
4.1.1. HAI as a First-Line Treatment without Systemic Therapy
In 2002, Tanaka et al. reported the clinical results for 11 never treated patients that underwent HAI with 5-FU, every 12 weeks using three different regimens, by means of a catheter and a port system percutaneously implanted. In addition, 36.3% of the enrolled patients had PVTT. Authors reported a DCR of 82% and a mean survival of 26 months. With regards to toxicity, 27% of patients experienced grade 34 AEs such as cholangitis and pancytopenia.
In 2009, Jarnagin et al. in a phase II study analyzed HAI FUDR at a dose of 0.16 mg/kg×20/pump flow rate, administered through a surgically implanted infusion pump in 34 patients . This clinical trial demonstrated a DRC of 88.2% higher in ICC patients rather than HCC ones and a median OS of 29.5 months. Grade 34 AEs, such as elevated bilirubin levels, abdominal pain, and diarrhea were reported in 20% of patients, while 6% experienced technique-related complications, such as infection and pump dislocation.
In 2013, Sinn et al. investigated biweekly HAI Oxaliplatin at the dosage of 85 mg/m2 in 120 min infusion and 5-FU in 37 patients. DCR was 64.9% and the median OS was 13.5 months. Thrombosis , dislocation , and infection were pump-related events. Grade 34 AEs including haematological cytopenia , increasing enzymes liver levels , and severe abdominal pain were reported.
4.1.2. HAI as a First-Line Treatment Combined with Systemic Therapy
Guidelines For Advanced Hcc
The Barcelona Clinic Liver Cancer staging system has become widely accepted in clinical practice and has been used in many clinical trials evaluating new drugs for HCC treatment . There are five HCC stages: very early stage 0, Child-Pugh A, single nodule < 2 cm diameter), early stage , intermediate stage , advanced stage , metastases ), and terminal stage .
According to the American Association for the Study of Liver Diseases practice guidelines for HCC management, sorafenib is now considered the first-line treatment for patients with advanced stage HCC with good liver function . Sorafenib inhibits the activity of several kinases, including Raf-1, B-Raf, vascular endothelial growth factor receptor 2, platelet-derived growth factor receptor, and c-Kit. It produces a clinically relevant increase in time to progression and length of survival, and its associated toxicity is easily managed without treatment-related mortality .
According to the most recent version of the clinical practice guidelines for HCC in Japan , and a study comparing HAIC with historical controls , HAIC may improve prognosis. At present, sorafenib remains the standard treatment for advanced HCC worldwide. Studies showing whether HAIC improves HCC prognosis relative to sorafenib are required in the future.
Don’t Miss: How Can You Spread Hepatitis
What Are The Side Effects And Benefits Of Arterial Chemotherapy Infusion
In reality, however, depending on the chemotherapeutic agent used, much of the drug does end up in the rest of the body. Therefore, selective intra-arterial chemotherapy can cause the usual systemic side effects. In addition, this treatment can result in some regional side effects, such as inflammation of the gallbladder, intestinal and stomach ulcers, and inflammation of the pancreas. HCC patients with advanced cirrhosis may develop liver failure after this treatment. So, what is the benefit of intra-arterial chemotherapy? The bottom line is that there is a greater likelihood of having a therapeutic effect on the cancer. Nevertheless, fewer than 50% of HCC patients will experience a reduction in tumor size.
What Is Arterial Chemotherapy Infusion And Chemoembolization Of Liver
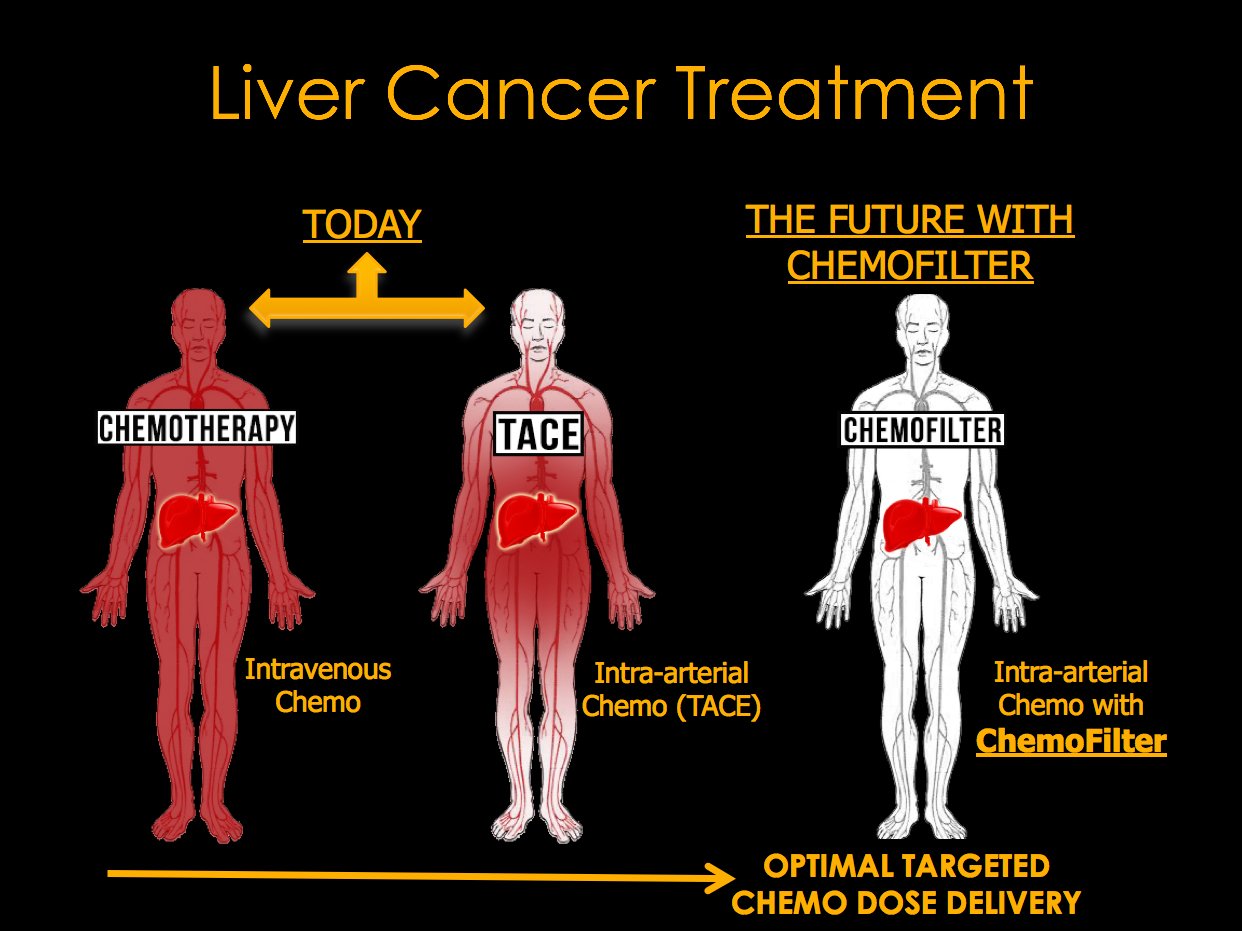
Arterial chemotherapy infusion of the liver and chemoembolization of the liver are similar procedures that are used for the treatment of cancers in the liver. In both procedures, chemotherapy is injected into the hepatic artery that supplies the liver tumor. The difference between the two procedures is that in chemoembolization, additional material is injected to block the small branches of the hepatic artery.
Read Also: Is There Immunization For Hepatitis C
Duke Surgeons Expertise With Hai Pumps
Drs. Lidsky and Allen and their Duke colleagues have implanted more than 60 pumps in people with liver metastases from colorectal cancer since they started offering the procedure in conjunction with whole-body chemotherapy in late 2018. In many cases, they also work alongside Duke surgical oncologist Sabino Zani, MD, who uses minimally invasive techniques, including robotic surgical approaches to implant the pump. The minimally invasive approach, which requires smaller incisions than traditional surgery, allows people to spend less time in the hospital, and recover faster.
Managing Toxicities And Adverse Effects
It is essential to balance our enthusiasm for liver-directed therapy with an awareness of the toxic effects of this treatment. Allen and colleagues found that 544 patients or 22 percent of patients treated at MSK from 1986 to 2001 experienced complications. The incidence of complications decreased with surgical experience. Most issues were salvageable, with 80 percent of pumps functioning for at least two years.
A review of drug-related toxic effects from HAI in 4,580 patients treated at MSK found gastrointestinal symptoms in 22 percent, hepatic toxic effects in 19 percent, and myelosuppression in 8 percent. HAI of floxuridine was mainly associated with hepatic enzyme elevations, which lead to biliary sclerosis in some people. Another MSK study found that 4.6 percent of patients undergoing HAI of floxuridine required a stent, and yet there was no difference in survival between those who received salvage treatment with a stent and dilation compared to patients without biliary complications. A further study among 50 randomized patients found a higher dose tolerance of floxuridine at five months and an increased response rate with concurrent administration of dexamethasone.
You May Like: What Are The Different Types Of Hepatitis
The Effectiveness Of Hai To Convert Initially Unresectable Colorectal Liver Metastases To Resection
The primary goal of multimodal treatment for initially unresectable colorectal liver metastases is to optimize response to make resection possible. Research has shown that patients whose disease can be converted to resection fare as well as those whose disease was resectable up front. ,
In the phase I trial at MSK noted above the study that examined the effectiveness of HAI in converting initially unresectable colorectal liver metastases to resectable or abatable disease in 49 patients conversion to resection was achieved in 47 percent of patients and up to as high as 57 percent for those who were chemotherapy naive. Despite a substantial burden of disease among these patients, and a strict definition of irresectability, the response rate was 76 percent. Forty-seven percent of patients achieved conversion to resection, which was the only factor independently associated with prolonged overall survival and progression-free survival. Three-year overall survival rates were an astonishing 80 percent for patients who underwent hepatectomy compared to only 26 percent for those whose disease remained inoperable.
Complications Associated With The Use Of Hai
Despite strong evidence suggesting that hai chemotherapy is effective at controlling crc liver metastases, enthusiasm and widespread adoption have been limited in part because of the significant complication rate associated with this therapy. Adverse events can be technical or toxicities related to chemotherapy .
Recommended Reading: What Doctor Treats Hepatitis C
Hepatic Arterial Infusion Pump Chemotherapy
HAIP chemotherapy has been developed by Dr. N.E. Kemeny, medical oncologist at MSKCC, and has been used for many years in the United States. HAIP chemotherapy involves continues infusion of intra-arterial chemotherapy using an implanted pump. Side-effects are uncommon since the chemotherapy has local effect in the liver. HAIP chemotherapy can be used solely and in combination with systemic chemotherapy. Studied patient groups include patients with resectable and non-resectable colorectal liver metastases and cholangiocarcinomas. The effect of HAIP chemotherapy is a reduction of the recurrence of the cancer in the liver and consequently improved survival in good quality of life. Secondly, HAIP chemotherapy can be used in patients with unresectable colorectal liver metastases. In this case, the aim is to prolong survival with good quality of life or, if possible, tumor shrinkage to a resectable state.
What Are The Benefits Of Tace
In one large study involving several institutions in Italy, chemoembolization did not seem to impact overall survival. Patients who did not undergo TACE lived as long as patients who received TACE, even though the tumors were more likely to shrink in size in patients who were treated. Does this mean that TACE or intra-arterial chemotherapy does not work? Maybe, maybe not.
Studies in Japan, however, have shown that TACE can downstage HCC. In other words, the tumors shrank enough to lower the stage of the cancer. From the practical point of view, shrinking the tumor creates the option for surgery in some of these patients. Otherwise, these patients had tumors that were not operable because of the initial large size of their tumors. More importantly, these same studies showed an improvement in survival in patients whose tumors became considerably smaller. In the U.S., trials are underway to see whether doing TACE before liver transplantation increases patient survival as compared to liver transplantation without TACE.
Also Check: How Long Is Hepatitis C Treatment
Hai Therapy Vs Systemic Chemotherapy For Unresectable Disease
HAI alone was initially compared to available systemic chemotherapies for first-line use for unresectable CRLM. Multiple prospective clinical trials published in the late 1980s to early 1990s comparing HAI with systemic chemotherapy demonstrated superior response rates of HAI therapy but did not show consistent improvements in OS . To add to the skepticism towards HAI therapy, a 2006 meta-analysis of randomized controlled trials comparing HAI and systemic chemotherapy in unresectable disease showed that there was no survival advantage to HAI alone . This analysis, however, had several limitations including, small number of patients from single institutions, old HAI chemotherapy, and allowance for cross over to HAI in patients who had initially failed systemic chemotherapy. To overcome these limitations, a multi-institutional prospective randomized clinical trial, Cancer And Leukemia Group B 9481, investigated response rate in patients receiving HAI FUDR compared to systemic 5-FU only, demonstrating significantly improved survival . In that study, 4070% of patients with unresectable hepatic liver metastases who underwent HAI therapy later developed extrahepatic disease thus, systemic chemotherapy combined with HAI was considered as a practical approach to control both intrahepatic and extrahepatic metastases .
Table 2Table 3
Hai For Unresectable Hcc
4.2.1. HAI as a First-Line Treatment without Systemic Therapy
In 2017, Kodama et al. conducted a retrospective cohort study with 68 patients treated with subcutaneous IFN + HAI 5-FU, by means of an implanted port-a-cath system, associated with 3 D-conformal radiotherapy delivered in 13 fractions ) and 40 patients treated with Sorafenib. The median OS and PFS were significantly longer in the HAI chemotherapy + RT group than in the Sorafenib group . The grade 3/4 AEs reported in the HAI chemotherapy + RT group were haematological events , increased AST or ALT , increased bilirubin level , diarrhea, general fatigue , and infection of the port system . In the Sorafenib group, the following grade 3/4 AEs were observed: Increased AST or ALT , increased bilirubin level , diarrhea and general fatigue , gastrointestinal bleeding , high blood pressure , and hand-foot syndrome .
In 2018, Choi et al. in a randomized, prospective, comparative trial evaluated HAI Cisplatin plus 5-FU , by means of an implanted port-a-cath system, every 34 weeks vs. Sorafenib . The ORR was 27.6% vs. 3.4% , OS was 14.9 months vs. 7.2 months and TTP was 4.4 months vs. 2.7 months in the HAI chemotherapy and Sorafenib groups, respectively. Severe AEs were hyperbilirubinemia , AST elevation , ascites , and catheter-related complications in the HAIC group and hyperbilirubinemia , hand-foot syndrome , and AST elevation in the Sorafenib group.
4.2.2. HAI as a First-Line Treatment Combined with Systemic Therapy
Recommended Reading: Cause Of Nausea And Vomiting In Hepatitis
Why Is The Chemotherapy Injected Into The Hepatic Artery
The normal liver gets its blood supply from two sources: the portal vein and the hepatic artery . Primary liver cancer, also known as hepatoma or hepatocellular carcinoma gets its blood exclusively from the hepatic artery. These techniques can also be used to treat secondary, or metastatic liver cancer, which is cancer that spread to the liver from other primary sites. These metastases also draw their blood supply from the hepatic arteries. This discussion will focus on primary liver cancer. Making use of this pattern of blood supply, investigators have delivered chemotherapy agents selectively through the hepatic artery directly to the HCC tumor. The theoretical advantage is that higher concentrations of the agents can be delivered to the cancer. The technique takes advantage of the concept of extraction: toxicity can be reduced by relying on the liver to extract or break down some of the chemotherapy after the tumor has been exposed to it before the chemotherapy gets through the liver into the systemic circulation.
How To Prepare For Hepatic Arterial Infusion
Your surgeon will give you specific guidance on how to prepare for your cancer surgery.
These guidelines may include the following:
- Complete any necessary preoperative testing at least a week before your surgery date. These might include blood and urine tests, a chest x-ray, an EKG, and others as needed.
- Stop taking aspirin, blood thinners, or anti-inflammatory drugs 10 days before your surgery. Your doctor will let you know if and when you should stop vitamins or other supplements.
- Stop eating and drinking at least eight hours before your surgery. Your doctor may let you take medicine with a sip of water the morning of surgery. If you have diabetes, ask whether you should take your diabetes medications on surgery day.
Also Check: Hepatitis C Antibody With Reflex To Hcv Rna
Rationale For Hepatic Artery Infusion Chemotherapy
There are approximately 150,000 new cases of colorectal carcinoma diagnosed annually in the United States . Approximately 25% of patients present with metastatic disease at the time of diagnosis and over 50% will develop metastasis to the liver at some point in their lifetime . Five-year overall survival in metastatic colorectal cancer confined to the liver is approximately 20%, although complete resection can increase 5-year survival to over 50% in selected series .
Liver resection provides the only chance for cure in patients with colorectal liver metastases , however, only 1520% of patients with CRC metastases confined to the liver are deemed resection candidates at presentation. Most patients with CRC die from metastatic disease, and two-thirds of CRC deaths are due to liver metastases .
HAI therapy has evolved in the last three decades with improved surgical techniques and discovery of new systemic agents. It is now an acceptable first line option in the United States for unresectable CRLM and a treatment option in the adjuvant setting however, it remains infrequently used. Reasons for this include lack of widespread expertise with HAI therapy, requirement of a multidisciplinary team to manage therapy, and more widespread use of systemic chemotherapy in first-line settings .
The Role Of Hepatic Artery Infusion
When colorectal cancer spreads, it frequently results in metastatic tumors in the liver. When possible, the liver tumors are surgically removed, and chemotherapy destroys the remaining cancer cells. Sometimes the tumors cant be removed because of their location or size, or the available treatments simply cant slow the disease. At this point, some people are told by their doctors that chemotherapy is their only option. Or worse, that nothing more can be done.
However, there is another option. It just hasnt been widely available because it requires specialized training in surgical and medical oncology.
Hepatic arterial infusion delivers chemotherapy through a pump thats implanted in the abdominal wall. We then use a catheter to deliver high doses of chemotherapy through the hepatic artery, which directly feeds metastatic tumors in the liver, said Peter Allen, MD, a Duke surgical oncologist. Even though the HAI pump delivers chemotherapy directly to the liver at concentrations that are hundreds of times higher than whole-body chemotherapy, it does not increase the side effects beyond those associated with whole-body chemotherapy.
If the liver tumors can be removed — doctors use the term resected — the pump may be implanted at the time of surgery and used to delay or prevent recurrence of cancer. If the liver tumors cannot be removed, the pump may be inserted to help control the tumor or, even better, shrink its size so it can be surgically removed in the future.
Recommended Reading: Help With Hepatitis C Treatment